The regulatory status of nicotinamide mononucleotide NMN has been one of the most closely watched stories in the supplement industry over the past several years, with the Food and Drug Administration (FDA) playing a central role in these decisions. If you’ve been confused about whether NMN supplements are legal, safe, or worth considering, you’re not alone. This guide breaks down exactly what happened, why the FDA reversed its position, and what you need to know as a consumer in 2026. As the landscape of NMN regulation continues to evolve, it’s important to note that the regulatory landscape for NMN varies globally, with different regions adopting their own approaches.
Quick Answers: NMN’s FDA Status in 2026
As of early 2026, beta nicotinamide mononucleotide is lawful for use in dietary supplements in the United States under current FDA rules. The FDA confirmed in September 2025 that NMN is lawful for use in dietary supplements in the United States. This represents a significant shift from the uncertainty that dominated the market between late 2022 and 2025.
The FDA’s reversal of its 2022 drug preclusion position in late 2025 followed sustained industry pressure through a formal citizen petition and litigation from the Natural Products Association. The FDA’s decision was based on a reassessment of the regulatory process, and the agency clarified its interpretation of the law regarding NMN’s status as a dietary ingredient. The FDA’s reversal means NMN can be reinstated as a permissible dietary ingredient based on evidence that it was marketed as a dietary supplement in the U.S. before the relevant drug investigation timeline. The FDA reverses NMN decision following pressure from the Natural Products Association, which filed a citizen petition and lawsuit challenging the FDA’s interpretation.
Here’s what’s important to understand: this was a regulatory classification question—drug versus supplement—not a ban based on safety concerns. NMN was never illegal to possess or use. The dispute centered entirely on whether companies could legally market NMN products as dietary supplements.
Consumers can now legally buy NMN supplements in the U.S. NMN listings are expected to be restored on Amazon and other major retailers following the FDA’s 2025 clarification, after previous removals due to regulatory uncertainty. However, even with the FDA’s reversal, NMN products face significant quality considerations and potential legal conflicts due to overlapping patents. Despite the FDA’s reversal, companies must still comply with all DSHEA rules, including labeling and manufacturing standards, and cannot make medical claims about NMN products. The FDA’s decision does not constitute an endorsement of NMN’s effectiveness for any health condition. The FDA’s clarification on NMN’s status is also expected to influence the global market for NMN supplements, as different countries have varying regulations.
The rest of this article unpacks why the US FDA changed course, what science and law were involved, and what practical steps consumers can take in 2026.
What Is NMN and Why It Matters
Nicotinamide mononucleotide is a naturally occurring compound derived from vitamin B3. It functions as a direct precursor to nicotinamide adenine dinucleotide (NAD+), a coenzyme found in every cell of your body that plays essential roles in energy production, DNA repair, and metabolic processes.
Here’s how the pathway works in simplified terms:
Your body takes various forms of vitamin B3 (niacin, niacinamide)
These get converted through several steps into NMN
NMN is then converted into NAD+
NAD+ powers hundreds of cellular reactions
Research from institutions including Harvard Medical School and Washington University has explored how NAD+ levels naturally decline with age, and whether supporting NAD+ production through precursors like NMN might influence aspects of healthy aging. This scientific interest sparked consumer demand for NMN supplements.
Between 2018 and 2024, the market for NMN grew rapidly, with the compound widely marketed as a “longevity” or “healthy aging” supplement. This popularity is precisely why its regulatory status attracted so much attention—and controversy.
It’s worth noting that scientific interest does not equal proven medical benefits. The enthusiasm around NMN is largely based on mechanisms observed in laboratory and animal studies, plus early human research, rather than definitive clinical endpoints for disease treatment.
How DSHEA and the Drug Preclusion Rule Shape NMN’s Fate
Understanding NMN’s regulatory journey requires knowing a bit about the Dietary Supplement Health and Education Act of 1994, commonly called DSHEA. This law created the framework for how dietary supplements are regulated in the United States—and it’s fundamentally different from how drugs are regulated.
Under DSHEA, supplements don’t require FDA approval before going to market. Instead, manufacturers are responsible for ensuring their products are safe and properly labeled. The Drug Administration FDA is the agency responsible for interpreting and enforcing these rules, and it can take action against products that are unsafe, misbranded, or make unauthorized disease claims.
However, DSHEA includes an important provision known as the drug preclusion clause. Here’s what it means in plain terms:
| Aspect | Drug Pathway | Dietary Supplement Pathway |
|---|---|---|
| Pre-market approval | Required | Not required |
| Clinical trial evidence | Extensive required | Safety evidence for new ingredients |
| Disease treatment claims | Permitted if approved | Not permitted |
| “First to market” consideration | Can exclude supplement use | Must predate drug investigation |
| The drug preclusion rule states that if a substance was first the subject of substantial clinical investigations as a new drug—and those investigations were made public—it generally cannot later be introduced as a dietary ingredient in supplements. This rule exists to protect pharmaceutical companies’ research investments while still allowing a robust supplement marketplace. |
For NMN, the critical question became: Was NMN legally marketed as a supplement before it became the subject of investigational new drug applications?
The timing of first lawful supplement marketing versus prior investigation as a drug became the core legal dispute that would take years to resolve.
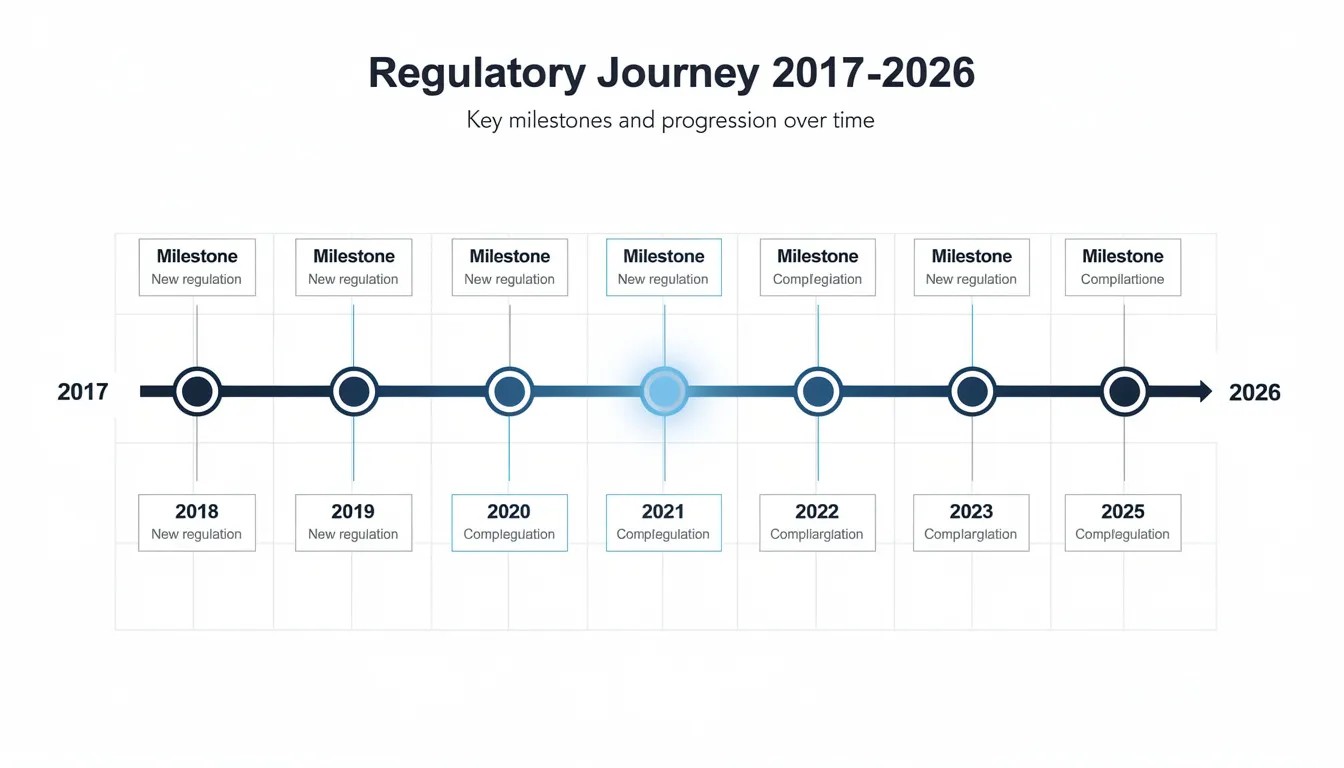
Timeline: From NDIN to Reclassification (2017–2026)
The regulatory journey of NMN spans nearly a decade. Here are the key milestones:
2017: Early U.S. Market Presence
NMN products begin appearing on supplement shelves in the United States
Multiple companies market NMN as a dietary ingredient without FDA objection
No formal regulatory guidance addresses NMN specifically
May 2022: FDA Acknowledgment
FDA issues a “good day letter” acknowledging New Dietary Ingredient Notifications for NMN
This seemingly positive signal suggested NMN’s supplement status was secure
November 2022: The Exclusion Letter
FDA issues a reversal letter excluding NMN from the dietary supplement definition
The agency invokes the drug preclusion clause, citing Metro International Biotech’s IND application for MIB-626
Retailers begin pulling NMN products amid confusion and fear of enforcement
Consumer access becomes severely limited despite NMN’s history in the market
March 2023: Industry Pushback Begins
The Natural Products Association files a citizen petition challenging FDA’s interpretation
NPA argues NMN was safely marketed as a supplement before substantial investigations began
Industry groups warn the decision could set a dangerous precedent for other lawful supplement ingredients
2023-2024: Legal Pressure Mounts
NPA files lawsuit against FDA
The June 2024 Supreme Court decision in Loper Bright Enterprises v. Raimondo ends Chevron deference
This ruling means courts no longer automatically defer to agency interpretations of statutes
FDA’s position becomes more vulnerable to legal challenge
Establishing NMN’s legitimacy required extraordinary investments, including legal actions and legislative efforts.
September 29, 2025: FDA Reverses Course
FDA issues two pivotal letters responding to the amended citizen petition
The agency concluded NMN is not excluded from the dietary supplement definition
FDA clarified that sufficient evidence showed NMN was marketed in the U.S. as a supplement prior to the drug authorization date
The reversal establishes that actual marketing—not necessarily full compliance—satisfies DSHEA requirements
NPA’s successful effort was a key milestone in securing regulatory clarity for NMN.
December 2025: NDI Notifications Reinstated
FDA sends letters to suppliers including SyncoZymes (Shanghai) Co. Ltd. and Inner Mongolia Kingdomway Pharmaceutical
These letters reinstate previously acknowledged dietary ingredient notifications
NMN can now be explicitly marketed as a supplement through compliant supply chains
Early 2026: Current Status
NMN is treated as a permissible dietary ingredient under DSHEA
Products return to the market, though quality and compliance vary
NDI notification requirements remain in place for manufacturers
Why the FDA Reclassified NMN: Legal, Scientific, and Policy Drivers
FDA’s decision to reverse its NMN exclusion didn’t happen because of a single study or political event. Instead, it resulted from a combination of legal pressure, documented marketing history, and evolving policy considerations, all within a dynamic regulatory landscape shaped by evolving laws and standards.
Legal Pressure and the Citizen Petition
NPA’s successful effort began with a formal citizen petition in March 2023, arguing that FDA had misapplied the drug preclusion rule. The petition documented evidence that NMN had been marketed as a dietary supplement in the United States well before Metro International Biotech’s drug applications became public.
The subsequent litigation forced FDA to reconsider its position. After the Loper Bright decision eliminated Chevron deference, agency interpretations faced heightened scrutiny from courts, creating additional legal risk for FDA if its NMN decision was challenged.
Marketing Evidence Prevailed
The FDA ultimately acknowledged that NMN’s pre-IND marketing history was sufficient to satisfy DSHEA requirements. The agency’s analysis determined that NMN met the statutory criteria because supplement sales predated the critical IND timeline.
Importantly, FDA confirmed that the standard is whether marketing actually occurred—not whether it was technically “lawfully marketed” with full compliance. This interpretation favored the supplement industry’s position. The FDA’s interpretation of DSHEA requirements was pivotal in the agency’s decision to reclassify NMN, shaping how regulatory guidelines were applied in this case.
No Safety Crisis
It’s worth emphasizing that no public safety crisis or new toxicity data drove FDA’s reversal. The shift was primarily about regulatory classification and consistent interpretation of DSHEA, similar to earlier debates over N-acetylcysteine (NAC).
Policy Concerns
FDA faced criticism from multiple directions:
Industry groups argued the broad application of drug preclusion was limiting consumer choice
Some congressional offices expressed concern about deterring investment in next generation ingredients
Consumer advocates worried about regulatory ambiguity undermining innovation
Pharmaceutical companies were seen as potentially using drug applications to block supplement competition
The NPA and other stakeholders successfully argued that FDA’s interpretation was chilling research and creating same uncertainty for other ingredients that congress intended DSHEA to protect.
What the Reclassification Does (and Does Not) Mean for Consumers
Consumers in 2026 have clearer legal access to NMN supplements in the U.S., but this clarity comes with important caveats.
What FDA’s Action Does Mean
NMN is lawful for use in dietary supplements
It is not automatically excluded by the drug preclusion rule
Companies can market NMN products if they comply with DSHEA requirements
Manufacturers must submit NDI notifications where required
Products must meet good manufacturing practices standards
What It Does Not Mean
FDA has not approved NMN as safe or effective for any medical condition
No company can legally market NMN as a treatment or cure for disease
Standard supplement disclaimers and rules still apply
This is not a safety endorsement—it’s a classification decision
Quality and purity are not guaranteed by FDA’s action
The reversal reduces legal disputes over sudden product removals based on drug preclusion, but it does not prevent FDA from acting against misbranded, adulterated, or poor-quality NMN products. Enforcement discretion remains with the agency for quality and claims violations.
Think of “lawful in supplements” as a minimum regulatory threshold, not a health guarantee. The FDA protects consumers from unsafe products and false claims, but it doesn’t pre-approve supplements the way it does drugs. This means individual responsibility for informed decision-making remains essential.
Science Snapshot: What Research Says About NMN and NAD+ (Without the Hype)
As of 2026, the scientific evidence for NMN includes:
Animal and Cell Studies
Extensive preclinical research exploring NAD+ metabolism
Studies in mice showing various effects on metabolic markers
Mechanistic work explaining how NMN is converted to NAD+
Human Trials
A growing number of small-scale studies (typically 25-100 participants)
Research from Japanese and U.S. institutions measuring NAD+ levels after NMN supplementation
Studies examining effects on biomarkers related to metabolism and physical function
Generally recognized mild to moderate side effect profiles at studied doses
What Studies Actually Measure
Most human studies focus on:
Changes in blood NAD+ or related metabolite levels
Biomarkers of metabolic health
Physical performance tests
Safety and tolerability
What they don’t measure (yet) with sufficient evidence:
Long-term health outcomes
Disease prevention or treatment
Lifespan extension in humans
The enthusiasm surrounding NMN is largely based on promising mechanisms and early data, not definitive clinical endpoints. No NAD+ precursor—including NMN—has been approved by FDA as a treatment for aging or any age-related disease.
If you’re evaluating NMN research, focus on what’s actually measured in peer-reviewed trials rather than marketing promises about reversing aging.
Quality, Purity, and Label Accuracy: Persistent NMN Market Risks
FDA’s reclassification solved a legal-category problem but did not automatically fix long-standing quality issues in the NMN supplement market.
The Quality Problem
Independent laboratory testing has repeatedly found significant problems with NMN products:
Many products don’t meet their labeled content claims
Some contain negligible amounts of actual NMN
Purity levels vary widely between manufacturers
Heavy metal contamination has been detected in some products
Why Quality Varies
Several factors contribute to inconsistent NMN quality:
| Factor | Impact |
|---|---|
| Complex synthesis | Requires specialized manufacturing expertise |
| Raw material variability | Most NMN is imported; quality varies by source |
| Limited oversight | Not all manufacturers follow rigorous testing protocols |
| Premarket notification requirements | Not all suppliers have filed or received NDI acknowledgments |
Red Flags to Watch For
Be cautious of NMN products that:
Lack batch-specific third-party testing certificates
Provide vague or missing ingredient sourcing information
Make unrealistic “miracle” health claims
Are priced unrealistically low for claimed purity or dosage
Don’t clearly identify beta-NMN content per serving
Come from companies with no verifiable manufacturing credentials
What to Look For
Quality indicators for NMN supplements include:
Clear labeling specifying beta-NMN content per serving
Published third-party lab results covering identity, purity (ideally >99%), heavy metals, and microbiological testing
Compliance with current Good Manufacturing Practices (cGMP)
Supplier with acknowledged NDI notification status
U.S.-based or clearly identified manufacturing facilities
Transparent company information and customer service

How NMN Compares to Other NAD+ Precursors (NR, Niacin, and More)
NMN is one of several NAD+ precursors available to consumers. Understanding how they compare can help inform your decisions.
| Aspect | NMN | NR (Nicotinamide Riboside) | Niacin (Nicotinic Acid) |
|---|---|---|---|
| Regulatory status (U.S.) | NDI with reinstated status | Established NDI + GRAS | Generally recognized as safe |
| Research maturity | Newer human data; growing | Longer human trial history | Decades of research |
| Common side effects | Generally mild in studies | Generally mild | Flushing common at higher doses |
| Typical studied doses | 250-1000mg daily | 250-500mg daily | Varies widely by form |
| Drug preclusion issues | Resolved (2025) | None | None |
Key Comparisons
NR (Nicotinamide Riboside) NR has a longer track record of human studies and established regulatory status, including both NDI acknowledgment and GRAS (generally recognized as safe) status. Some researchers consider it a more stable alternative with robust research backing.
Mechanistic Relationship Some studies suggest NMN is converted to NR or related intermediates before entering cells. Both NR and NMN ultimately feed into the same NAD+ biosynthesis pathways, raising questions about whether one offers meaningful advantages over the other.
Niacin and Niacinamide Traditional B3 vitamins remain the most studied and affordable NAD+ precursors, though they work through slightly different pathways and (in niacin’s case) often cause flushing side effects.
Avoid getting caught up in “either-or” marketing hype. Consider factors like evidence quality, personal tolerance, cost, and advice from health professionals when evaluating any NAD+ precursor.
Global Snapshot: NMN Regulation Beyond the United States
NMN’s regulatory story extends beyond U.S. borders, and understanding the global landscape helps contextualize product availability and quality standards. The regulatory landscape for NMN supplements varies significantly across regions, with the U.S. allowing NMN, the EU requiring Novel Food authorization, and China imposing strict regulations.
European Union
The EU treats NMN as a Novel Food ingredient requiring pre-market authorization. On July 25, 2025, the European Food Safety Authority (EFSA) launched a public consultation on NMN (application PC-1537 from Shanghai Shangke Biotechnology). As of early 2026, no general authorization has been granted, and conditions for any approved use would likely be strict.
China
China serves as a major production hub for NMN raw materials. However, domestic sale of NMN as a food or supplement ingredient remains restricted. NMN is primarily regulated there as a cosmetic raw material or accessed through narrow personal import channels.
Other Markets
Japan: Has allowed NMN supplements with fewer specific ingredient barriers
Australia: Maintains tighter control with case-by-case ingredient evaluation
Canada: Regulates supplements as Natural Health Products with pre-market review requirements
What This Means for Consumers
This regulatory patchwork means:
Manufacturing standards vary by country of origin
Permitted claims differ across markets
Consumer protections are inconsistent internationally
Products purchased online may come from varying regulatory environments
Prioritize products that clearly state where they are made, how they are tested, and how they comply with relevant regulations—especially when purchasing cross-border or from international online retailers.
Practical Tips for Consumers Considering NMN in 2026
NMN is now lawful as a dietary supplement in the U.S., but decisions about use should be thoughtful, evidence-informed, and tailored to your individual circumstances.
Before You Start
Consult a healthcare professional if you are pregnant, nursing, managing chronic conditions, taking prescription medications, or considering doses higher than those typically studied
Set realistic expectations—NMN is not a proven treatment for any disease
Understand that supplement category status means minimal pre-market oversight
What to Review Before Purchasing
Use this checklist when evaluating NMN products:
[ ] Ingredient list clearly identifies beta-NMN
[ ] Dosage per serving aligns with published study ranges (typically 250-1000mg)
[ ] Third-party testing certificates are available and current
[ ] Manufacturer provides transparent company information
[ ] No disease-treatment claims appear on labeling or marketing
[ ] Supplier has acknowledged NDI notification (can verify via FDA portal)
[ ] Product is manufactured in cGMP-certified facilities
Starting Safely
If you decide to try NMN:
Start with the lowest practical dose on the label
Monitor for any side effects
Work with a clinician rather than self-escalating rapidly
Avoid combining multiple NAD+ boosters without professional guidance
Keep records of what you take and any effects you notice
Don’t Forget the Fundamentals
Track objective lifestyle factors alongside any supplement use:
Sleep quality and duration
Dietary patterns
Physical activity levels
Stress management practices
These fundamentals have strong evidence for supporting long-term health. NMN, if used, should complement—not replace—core health habits.

Looking Ahead: What to Watch for in NMN Research and Policy
NMN’s story is still unfolding. Both regulatory guidance and scientific evidence will likely evolve over the coming years.
Upcoming Research
Additional human clinical trials are underway or planned examining:
Long-term safety profiles
Optimal dosing strategies
Effects on specific health-related biomarkers
Comparisons with other NAD+ precursors
Results from these studies could influence future administrations’ regulatory attitudes toward NMN and similar ingredients.
Potential Policy Developments
Several policy changes could affect NMN’s future:
Congressional action: Some lawmakers have expressed interest in clarifying or amending the drug preclusion clause in DSHEA, which could provide more stability for NMN and similar ingredients, though absent congressional action, current interpretations stand
Targeted legislative fix: Industry groups continue pushing for reforms that would prevent future regulatory ambiguity
Enforcement focus: FDA may increase attention on labeling, manufacturing quality, and claim substantiation for NMN and other longevity supplements
Persistent Uncertainties
Despite FDA’s reversal, some legal risk remains. The post-Loper Bright environment means agency interpretations can be challenged more easily in court. Private litigation or subsequent litigation over DSHEA’s provisions could reopen debates about which ingredients qualify for supplement status.
Future administrations may also interpret these rules differently, creating ongoing regulatory hurdles for supplement businesses operating in this space.
Staying Informed
Look for updates from authoritative sources:
FDA communications and guidance documents
Peer-reviewed journals (not just press releases)
Reputable scientific organizations
Consumer advocacy groups focused on supplement quality
Avoid relying solely on commercial marketing materials, which often overstate benefits and minimize uncertainties.
The NMN regulatory saga from 2022 to 2026 demonstrates how supplement law, pharmaceutical development, and consumer demand can intersect in complex ways. NPA’s successful effort to reverse FDA’s exclusion provides regulatory clarity for now, but the broader questions about how DSHEA handles dual-use ingredients remain unresolved.
For consumers, the practical takeaway is straightforward: NMN is lawful, but lawfulness is just the starting point. Quality matters. Science is still evolving. And the fundamentals of healthy living—sleep, nutrition, movement, stress management—remain your most evidence-backed investments.
Stay curious, stay skeptical of hype, and keep the conversation going with healthcare professionals who know your individual situation. That approach will serve you well whether you’re considering NMN or any other emerging supplement in the years ahead.
Further Reading
Explore more articles related to this topic:
- Is NMN Legal? Navigating the FDA and International Regulations in 2026
- Is NMN Safe for Your Liver? Analyzing the Latest 2026 Hepatotoxicity Data
- NMN: Everything You Need to Know in 2026
- Cheap NMN vs. Premium NMN: Why 99% Purity Matters for Long-Term Safety
- The 2026 Runner’s Guide: How NMN Impacts VO2 Max and Aerobic Capacity




