If you’ve been following longevity science, you’ve probably noticed NAD+ popping up everywhere—from research journals to supplement shelves. But understanding how NAD+ actually works across your entire body has been a moving target for scientists. Enter NAD World 3.0, the latest theoretical framework that attempts to explain how this essential molecule coordinates aging and longevity at a systems level.
This isn’t just another buzzword. It represents over 15 years of research culminating in a more complete picture of how your body maintains cellular health as you age. Let’s break down what this framework actually means and why it matters for anyone interested in healthy aging.

Quick Explanation: What NAD World 3.0 Actually Is
NAD World 3.0 is a scientific framework that describes how nicotinamide adenine dinucleotide (NAD+) metabolism coordinates aging and longevity in mammals. Think of it as a map showing how different organs communicate to maintain your body’s NAD+ supply—the molecular currency that powers everything from DNA repair to energy production.
The term was formally introduced in 2025 by Shin-Ichiro Imai and his research team at Washington University in St. Louis, with their work published in npj Aging. This framework builds directly on earlier iterations: the original NAD World concept from 2009 and NAD World 2.0 from 2016.
In plain English, NAD World 3.0 describes how four key tissues work together:
Hypothalamus – the brain’s command center
Skeletal muscle – the metabolic workhorse
Adipose tissue – the fat-based modulator
Small intestine – the newly recognized signal generator
These tissues communicate using nicotinamide mononucleotide (NMN), the NMN transporter Slc12a8, and extracellular NAMPT (eNAMPT) to maintain NAD+ balance throughout your body.
It’s important to clarify that this is a theoretical model supported by animal studies and early human research—not a clinical treatment guideline. At Vitruvin, we focus on this science because many longevity supplements, including NMN and resveratrol, intersect directly with NAD+ biology and cellular health. Understanding the underlying framework helps contextualize what these compounds might actually be doing in your body.
Background: How the NAD World Concept Evolved (2009 → 2016 → 2025)
Science rarely arrives fully formed. The NAD World concept has evolved through three distinct phases over 15 years, each adding layers of understanding about how NAD+ influences aging and longevity.
NAD World (2009): The Foundation
The original NAD World concept established several foundational ideas:
NAD+ and SIRT1 connection – SIRT1, an NAD+-dependent deacetylase enzyme, plays a critical role in DNA repair, metabolic regulation, stress resistance, and circadian rhythm modulation
NAMPT as the rate limiting enzyme – This enzyme synthesizes NAD+ from nicotinamide via the salvage pathway, making it essential for maintaining cellular NAD+ levels
Systemic aging control – Rather than viewing aging as random cellular wear-and-tear, Imai proposed that NAD+ and SIRT1 function as central regulators of longevity at the cellular level
Circadian rhythm integration – The framework linked NAD+ metabolism to biological rhythm and daily metabolic cycles
This initial model positioned NAD+ as more than just a metabolic cofactor—it framed the molecule as a potential master regulator of aging processes.
NAD World 2.0 (2016): Adding Inter-Tissue Communication
Seven years of additional research led to significant updates:
Three key tissues identified – The hypothalamus (control center), skeletal muscle (metabolic mediator), and adipose tissue (systemic modulator) emerged as core players
Extracellular NAMPT discovery – Scientists found that NAMPT isn’t just intracellular; a secreted form called eNAMPT acts as a messenger between tissues
NMN as a circulating signal – The framework proposed that NMN travels between organs to support NAD+ biosynthesis in various tissues
Inter-tissue coordination – Rather than operating in isolation, these organs form a coordinated network maintaining NAD+ homeostasis
What Remained Unknown Before 2025
Despite these advances, several gaps persisted:
How does NMN actually enter cells?
How is NAD+ controlled in specific tissues?
How does the body orchestrate fast versus slow NAD+ regulation?
What explains the spatial and temporal dynamics of NAD+ distribution?
These unanswered questions set the stage for NAD World 3.0, which adds the small intestine as a fourth pillar and introduces the NMN transporter Slc12a8 as a key mechanism for key inter tissue communication.
Core Idea of NAD World 3.0
At its heart, NAD World 3.0 proposes that your body maintains NAD+ through a systemic regulatory network—a multi-layered communication model that explains how NAD+ is coordinated in space (between organs) and time (minutes versus days).
The framework centers on two main molecular players:
| Molecule | Function |
|---|---|
| NMN (nicotinamide mononucleotide) | A direct NAD+ precursor that can be rapidly transported into cells |
| eNAMPT (extracellular NAMPT) | A secreted enzyme released in vesicles that helps generate NMN at distant sites |
| The small intestine emerges as a “signal generator” in this model, thanks to the NMN transporter Slc12a8. This transporter controls rapid NMN uptake from the gut into cells, enabling quick adjustments to systemic NAD+ availability. |
NAD World 3.0 proposes that aging isn’t a problem in just one organ—it’s the progressive breakdown of NAD+-linked multi layered feedback loops between brain, muscle, fat tissue, and intestine.
This framework is primarily based on mouse research, including Imai’s 2025 publication in npj Aging. Supporting evidence comes from cellular models and early clinical research on NAD+ precursors like NMN and nicotinamide riboside.
The Four Key Tissues in NAD World 3.0
The NAD World 3.0 framework identifies four key tissues that form an interconnected network for maintaining NAD+ homeostasis throughout the body. Each plays a distinct role:
| Tissue | Role in NAD World 3.0 |
|---|---|
| Hypothalamus | Command center |
| Skeletal muscle | Effector/mediator |
| Adipose tissue | Modulator |
| Small intestine | Signal generator |
| These aren’t isolated players—they communicate through NMN, eNAMPT-containing extracellular vesicles, and sympathetic nervous system signals. Let’s examine each one. |
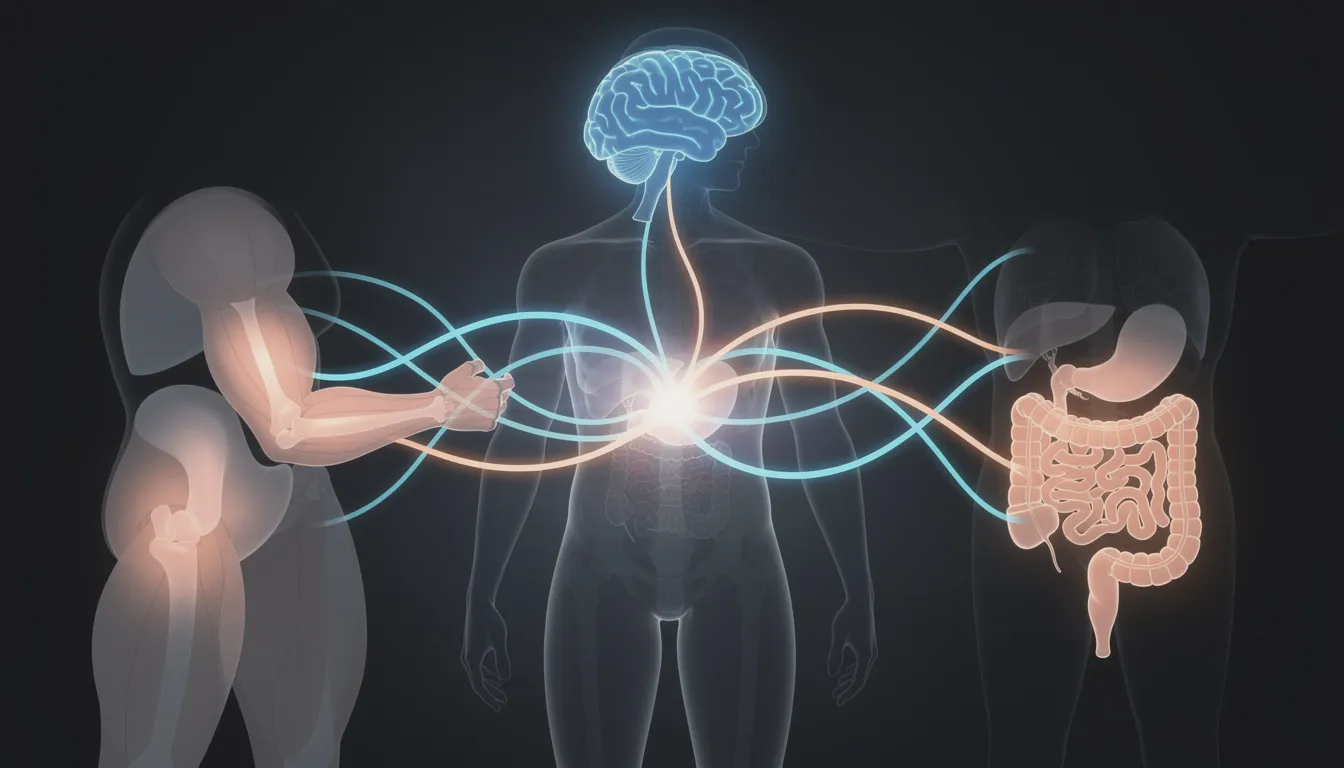
The Hypothalamus: Central Control of Aging Signals
The hypothalamus is a small but powerful brain region that regulates body temperature, energy balance, circadian rhythm, and hormonal axes. In the NAD World framework, it functions as the control center that sends signals to peripheral tissues.
Key findings from research include:
NAD+ levels in hypothalamic neurons appear to influence lifespan and healthspan in mice, partly by modulating SIRT1 activity and sympathetic nervous output
Age-related hypothalamic NAD+ decline is associated with disrupted biological rhythm, altered hormonal signaling, and systemic frailty in animal models
eNAMPT vesicles from adipose tissue can raise hypothalamic NAD+ and support neuronal function in preclinical studies
The hypothalamus essentially acts as an orchestrator, receiving NAD+-related signals from fat tissue and dispatching instructions to skeletal muscle and other organs. When hypothalamic NAD+ levels fall—as they appear to with age—this coordination weakens, potentially contributing to age related decline across multiple tissues.
Skeletal Muscle: Metabolic Workhorse and Longevity Mediator
Skeletal muscle is a major consumer of NAD+ due to its high mitochondrial energy demand. Every time you move, your muscles are burning through ATP—and NAD+ is essential for that energy production process.
Research has linked age-related NAD+ decline in muscle to several problems:
Reduced mitochondrial function and lower ATP generation
Sarcopenia (loss of muscle mass and strength)
Decreased metabolic flexibility
Impaired insulin sensitivity
In the NAD World framework, the hypothalamus sends sympathetic nerve signals to muscle, and these signals are influenced by hypothalamic NAD+ status. Clinical work from small NMN trials published around 2021-2023 has explored NMN’s effects on skeletal muscle insulin sensitivity and metabolic markers, though long-term functional outcomes remain under active investigation.
The takeaway: skeletal muscle serves as the “mediator” in NAD World 3.0, translating central NAD+ signals into physical capacity, mobility, and metabolic flexibility. This positioning helps explain why age related senescence in muscle tissue can have such widespread effects on overall health.
Adipose Tissue: eNAMPT Source and Metabolic Modulator
Adipose tissue has undergone a reputation rehabilitation in recent decades. Scientists now recognize it not just as fat storage but as an endocrine organ releasing hormones and signaling proteins that affect the entire body.
In NAD World 3.0, adipose tissue serves as a major source of eNAMPT containing extracellular vesicles. Here’s how it works:
Fat cells package active NAMPT enzyme into extracellular vesicles (EVs)
These eNAMPT vesicles circulate through the bloodstream
They deliver NAMPT to distant tissues like the hypothalamus and hippocampus
Recipient tissues use the delivered NAMPT to produce NMN and subsequently NAD+
An important distinction: eNAMPT in EVs exists as an enzymatically active dimer that supports NMN production, whereas monomeric eNAMPT has been associated with inflammation in some contexts.
Mouse studies from Imai’s lab demonstrate that mice lacking adipose eNAMPT exhibit accelerated NAD+ decline and premature aging phenotypes—effects that can be reversed by EV infusions. This highlights adipose tissue’s role as a “modulator,” adjusting NAD+ support to vulnerable organs based on metabolic and sympathetic nervous signals.
Small Intestine: NMN Transport and the New “Signal Generator”
The small intestine represents the newly recognized “fourth pillar” in NAD World 3.0, primarily due to the discovery of the NMN transporter Slc12a8.
Before this discovery, scientists weren’t entirely sure how NMN entered cells after oral consumption. The identification of Slc12a8 as a specific NMN transport mechanism answered this question—at least in mice.
Key findings about the small intestine’s role:
Slc12a8 is highly expressed in the small intestinal epithelium
Genetic Slc12a8 deficiency reduces NMN uptake and intestinal NAD+
Slc12a8 overexpression increases NAD+ levels in intestinal tissue
Age-related upregulation of Slc12a8 in aged intestines suggests a compensatory response to NAD+ decline
This positions the small intestine as a “signal generator” that can quickly adjust systemic NAD+ availability in response to dietary inputs and age-related changes. When you consume NMN supplements or NMN-containing foods, the small intestine is where the action begins.
The gut microbiome may also contribute NMN and related metabolites, hinting at a future research connection between microbiota, Slc12a8, and systemic NAD+ homeostasis. This represents an exciting frontier in aging research.
Molecular Players: NAD⁺, NMN, NAMPT, and eNAMPT
Understanding NAD World 3.0 requires familiarity with its key molecular players. Here’s a breakdown of each component and how they interact in the NAD+ salvage pathway.
The NAD+ Salvage Pathway (Simplified)
Nicotinamide → (NAMPT/eNAMPT) → NMN → (NMNAT) → NAD+
↑ ↑
Rate-limiting step Final conversion
NAD+ (Nicotinamide Adenine Dinucleotide)
NAD+ is an essential coenzyme involved in hundreds of cellular reactions:
Redox reactions for energy metabolism
Mitochondrial ATP production
DNA repair mechanisms
Sirtuin activation (including SIRT1)
Epigenetic regulation
Research consistently shows that NAD levels generally decline with age in multiple tissues. Studies in rodents suggest 50-70% NAD+ drops across various tissues by middle age.
NMN (Nicotinamide Mononucleotide)
NMN is a direct precursor to NAD+ produced from nicotinamide via NAMPT. It represents the final step before NAD+ synthesis.
Oral NMN has been tested in multiple human studies for safety and NAD+ effects
NMN supplementation is being actively researched for its potential health benefits
NMN levels appear to fall proportionally with NAD+ during aging
NAMPT (Intracellular)
NAMPT is the rate limiting enzyme in the NAD+ salvage pathway. It converts nicotinamide to NMN inside cells, making it essential for maintaining intracellular NAD+ levels.
eNAMPT (Extracellular NAMPT)
eNAMPT is the secreted form of NAMPT that helps generate NMN outside cells. In NAD World 3.0, eNAMPT often travels within extracellular vesicles, acting like a hormone-like messenger regulating NAD+ production at distant sites such as the hypothalamus.
Research indicates that eNAMPT secretion diminishes by up to 40% in adipose tissue with age, potentially explaining some aspects of systemic NAD+ decline.
While boosting NAD+ with precursors (NMN, NR, etc.) is actively studied, long-term health outcomes, optimal dosing, and population-specific responses are still being established through clinical studies.

Slc12a8: The NMN Transporter at the Heart of NAD World 3.0
Slc12a8 is a sodium-dependent transporter protein identified as a specific NMN carrier in mouse small intestine and some brain regions. Its discovery addressed a longstanding question in NAD+ research: how does NMN actually get into cells?
Experimental Evidence
Studies have demonstrated several key findings about this NMN transporter:
| Intervention | Outcome |
|---|---|
| Genetic reduction of Slc12a8 | Lower NMN uptake and reduced NAD+ |
| Slc12a8 restoration | Rescued NMN transport and NAD+ synthesis |
| Small intestine Slc12a8 knockout | Tissue-specific NAD+ deficits |
Age-Related Changes
Interestingly, Slc12a8 expression is upregulated in aged intestinal tissue. This suggests the body may attempt to counter systemic NAD+ decline by increasing NMN absorption capacity—a compensatory response that may become insufficient as aging progresses.
Slc12a8 is also expressed in the lateral hypothalamus, where its age-related decline has been linked in aged mice to sarcopenia and frailty due to disrupted hypothalamus-muscle communication.
Human Relevance
The human relevance of Slc12a8-mediated NMN transport remains an active area of investigation. While the transporter exists in humans, expression patterns and functional significance may differ from mouse models. Previous studies provide a foundation, but extrapolating mouse findings to human clinical outcomes requires caution.
Fast vs. Slow NAD⁺ Control: Two Interlocking Systems
One of NAD World 3.0’s key insights is distinguishing between “fast” and “slow” components of NAD+ regulation. Your body uses different mechanisms for different time scales.
The Fast Lane: Minutes to Hours
The fast lane operates through Slc12a8-mediated NMN transport:
Location: Primarily small intestine and select tissues
Mechanism: Direct NMN uptake through Slc12a8 transporter
Speed: Minutes to hours
Purpose: Meeting acute energy or repair demands
Activation: SIRT1 and other sirtuins activated rapidly
When you consume NMN supplements or NMN-containing foods, this pathway enables relatively rapid increases in intracellular NMN and NAD+. Mouse models show that timed NMN availability can sustain muscle NAD+ for up to 24 hours post-dose via Slc12a8.
The Slow Lane: Hours to Days
The slow lane operates through eNAMPT-containing extracellular vesicles:
Source: Primarily adipose tissue
Mechanism: EV-packaged eNAMPT travels through circulation
Speed: Hours to days
Purpose: Sustaining baseline NAD+ in target tissues
Targets: Hypothalamus, hippocampus, and other vulnerable organs
Think of it this way: the fast lane is like a quick top-up for immediate needs, while the slow lane functions as a long-term savings account for NAD+. Both systems converge on the same outcome—maintaining NAD+ homeostasis to support mitochondrial function, DNA repair, and epigenetic regulation.
Why This Matters
These two systems operate on different time scales but work together to maintain NAD+ levels across various tissues. When either system falters—whether through reduced Slc12a8 expression or diminished eNAMPT-EV production—the body’s ability to maintain NAD+ balance becomes compromised.
Understanding this dual-pathway system helps explain why single interventions might have limited effects. Supporting both fast and slow pathways may be more effective than targeting just one.
Aging Through the Lens of NAD World 3.0
NAD World 3.0 views aging as a progressive loss of NAD+ “signaling robustness” across the four-tissue network. Rather than seeing aging as inevitable cellular breakdown, this framework emphasizes the deterioration of communication between organs.
The Cascade of NAD+ Decline
Systemic NAD+ decline has been associated with multiple aging-related changes:
| Process | Connection to NAD+ |
|---|---|
| Mitochondrial dysfunction | NAD+ required for electron transport chain |
| Inflammaging | Inflammatory cytokines downregulate NAMPT |
| Altered epigenetics | NAD+-dependent sirtuins influence chromatin |
| DNA damage accumulation | NAD+ powers PARP enzymes for repair |
| Cellular senescence | Senescent cells have disrupted NAD+ metabolism |
| Metabolic dysfunction | NAD+ essential for metabolic flexibility |
| Inflammatory cytokines can downregulate NAMPT while upregulating NAD-consuming enzymes like CD38, compounding NAD+ loss and impairing sirtuin activity in aging tissues. |
NAD World 3.0’s Integrated View
The framework integrates these observations into a systems-level model:
Weakened hypothalamic NAD+ disrupts central coordination
Reduced eNAMPT-EV output from adipose tissue limits slow-lane support
Altered Slc12a8 expression impairs fast-lane NMN uptake in specific tissues
Impaired communication to skeletal muscle contributes to sarcopenia and frailty
In young mice, these systems function robustly with strong connections between all four pillars. With age, these connections weaken, feedback loops become less responsive, and the body’s ability to maintain NAD+ homeostasis degrades.
Context Matters
While this systems view is compelling, it’s important to recognize that mammalian aging is multifactorial. NAD+ represents one important layer among genetics, lifestyle factors, environmental exposures, and other molecular pathways. Viewing NAD World 3.0 as one piece of a larger puzzle rather than the complete picture maintains appropriate scientific humility.
Implications for Longevity Research and NAD⁺-Focused Interventions
NAD World 3.0 highlights potential therapeutic targets—but does not by itself validate specific therapies for humans. Here’s a balanced overview of where the research stands.
Current Human Data
Small clinical trials of NMN and nicotinamide riboside have shown:
Increases in whole blood and blood samples NAD+ levels
Some improvements in metabolic markers (e.g., insulin sensitivity, lipid profiles)
No major safety concerns over intervention periods of weeks to months
Significant increase in NAD+ levels in study participants receiving supplementation
However, these trials often involve modest sample sizes and limited duration. Statistical analysis shows promise, but achieving statistical significance for long-term health outcomes requires larger, longer studies. Some research has shown statistically significant difference in biomarkers, but functional outcomes remain under investigation.
Future Research Directions
Based on NAD World 3.0, future approaches may explore:
Modulating eNAMPT-EV release pharmacologically or through lifestyle
Protecting hypothalamic NAD+ through targeted interventions
Influencing gut-brain-muscle communication via diet, exercise, and microbiome-targeted strategies
Developing Slc12a8 modulators for enhanced NMN uptake
The Bigger Picture
NAD+ precursors represent one tool within a broader longevity toolbox. The clinical evidence supports their safety and ability to raise NAD+ levels, but effects on biological age and age related disease prevention require more research.
Other evidence-based strategies remain essential:
Regular physical activity
Quality sleep and circadian alignment
Balanced nutrition
Stress management
Social connection
Viewing NAD+-supportive strategies as ways to support general cellular and metabolic health—rather than as disease treatments—reflects current clinical evidence appropriately.
Tips for Readers Interested in Supporting Their NAD⁺ System
Based on current research, here are practical, non-medical lifestyle suggestions that may support NAD+-related pathways.
Physical Activity
Regular exercise, especially endurance and resistance training, is associated with:
Healthier mitochondrial function
Upregulated NAD+-related gene expression
Improved metabolic flexibility
Better muscle NAD+ status in animal models
Aim for a combination of aerobic activity and strength training. Both modalities appear to support different aspects of cellular metabolism.
Sleep and Circadian Alignment
NAD+ metabolism is closely linked to circadian rhythm. Consider:
Maintaining consistent sleep-wake times
Getting morning light exposure
Limiting blue light and screen time in evening hours
Avoiding large meals close to bedtime
These habits may help support NAD+-linked clock genes and metabolic regulation, potentially improving your energy level and overall metabolic health.
Nutrition
A nutrient-dense diet supports NAD+ pathways through multiple mechanisms:
Adequate protein provides amino acid precursors
Polyphenol-rich foods (berries, green tea, grapes containing resveratrol) may support sirtuin activity
Avoiding chronic overnutrition reduces metabolic stress
Fasting glucose optimization through balanced eating patterns
Some research suggests that time-restricted eating patterns aligned with circadian rhythms may support metabolic health, though more research is needed.
Considering NAD+ Precursors
Some individuals choose to discuss NAD+ precursors with their healthcare professionals. If considering NMN supplements or similar products:
Review baseline measurements of your current health status
Discuss with a qualified healthcare provider, especially if you have existing conditions or take medications
Look for products that have been independently tested for purity
Consider one capsule or starting doses recommended by manufacturers
Monitor for any significant change in how you feel
Supplements are not substitutes for medical treatment. Any decisions about supplement use should be made in consultation with qualified health professionals who can provide personalized guidance upon reasonable request.

Visual Aids and Suggested Images
Understanding NAD World 3.0 benefits from visual representations. Here are descriptions of helpful diagrams:
NAD World 3.0 Overview Diagram
A figure showing the four tissues (hypothalamus, skeletal muscle, adipose tissue, small intestine) arranged in a network pattern with:
Arrows representing NMN flow between tissues
Dashed lines showing eNAMPT-EV signaling
Solid lines indicating sympathetic nervous connections
Labels for each tissue’s role (command, mediator, modulator, signal generator)
NAD+ Salvage Pathway Graphic
A simplified pathway showing:
Nicotinamide as the starting point
NAMPT/eNAMPT catalyzing conversion to NMN
NMNAT completing conversion to NAD+
Slc12a8’s role in NMN uptake highlighted
Clear labels for each enzyme and intermediate
Fast vs. Slow Timeline
A dual-track visualization showing:
Upper track: Slc12a8-mediated rapid response (minutes to hours)
Lower track: eNAMPT-EV support (hours to days)
Meeting point showing convergence on NAD+ homeostasis
Timeline markers indicating relative speeds
Signaling Robustness Visualization
A conceptual image showing:
Youth: Dense, strong connections between four tissues with vibrant signal flows
Aging: Gradually thinning connections, reduced signal strength
Representing how reduced NAD+ impairs feedback loops over time
Research Status, Open Questions, and How to Read the Science Carefully
NAD World 3.0 represents an evolving research framework, not a finished map of aging. Treating it as a complete picture would be premature.
Open Questions
Several key questions remain unanswered:
Translation to humans: How closely do mouse findings apply to human physiology?
eNAMPT-EV manipulation: Can we safely influence eNAMPT-EV release in humans?
Human Slc12a8: What is the full role of Slc12a8 in human tissues?
Long-term outcomes: What are the effects of NAD+ precursor supplementation over years, not months?
Individual variation: Why do some people respond differently to NAD+ interventions?
A significant limitation of current research is the reliance on animal models. While mice provide valuable insights, human aging involves additional complexity.
Reading Studies Critically
When evaluating NAD+ research, consider:
| Factor | What to Look For |
|---|---|
| Sample size | Larger studies provide more reliable data |
| Duration | Intervention period length affects clinical relevance |
| Population | Animal vs. human; age and health status of study participants |
| Outcomes | Mechanistic (NAD+ levels) vs. clinical (function, disease) |
| Repeated measures | Multiple baseline measurements and timepoints strengthen conclusions |
| Blood collection methods | Standardized whole blood or blood samples processing |
| Statistical analysis | Appropriate methods and reported statistical significance |
| Pay attention to conflicts of interest and funding sources. Industry-funded trials aren’t automatically invalid, but transparency matters. |
Vitruvin’s Approach
From our perspective, the goal is to align with high-quality, peer-reviewed evidence while avoiding overstating what current data can support. Cell Rep and other reputable journals have significantly advanced our understanding through publications like those cited in NAD World 3.0, but healthy skepticism remains appropriate.
We believe in providing helpful content that empowers readers to make informed decisions rather than promotional claims that oversimplify complex science.
Summary and Take-Home Messages
NAD World 3.0 represents a significant evolution in our understanding of how NAD+ influences aging and longevity. Here are the key takeaways:
Systems-level framework: NAD World 3.0 frames aging as a breakdown of NAD+-dependent communication between hypothalamus, skeletal muscle, adipose tissue, and small intestine—the three key tissues from earlier models plus the newly recognized intestinal pillar
Central molecular players: NMN, eNAMPT, and the NMN transporter Slc12a8 coordinate NAD+ supply over different time scales—fast (minutes to hours) and slow (hours to days)
Beyond single-organ aging: The model shifts perspective from isolated cellular damage to progressive deterioration of inter-organ signaling networks
Research foundation: While primarily based on mouse research, the framework integrates findings from multiple tissues, physiological processes, and related human studies
Lifestyle foundations remain essential: Healthy aging strategies including exercise, sleep, nutrition, and stress management support NAD+-related pathways regardless of supplementation choices
Ongoing research: The next decade will clarify how best to translate NAD World 3.0 concepts into safe, effective strategies to support cellular health and healthy longevity in humans
The original NAD World concept in 2009 proposed that NAD+ plays a critical role in coordinating mammalian aging. Fifteen years later, NAD World 3.0 has significantly advanced this vision by mapping the specific tissues, transporters, and signaling mechanisms involved.
Whether you’re considering NMN supplementation, optimizing your lifestyle for longevity, or simply curious about cutting-edge aging research, understanding NAD World 3.0 provides valuable context. The science continues to evolve, and staying informed helps you make better decisions about your own health journey.
For those interested in exploring NAD+ science further, consulting peer-reviewed sources and discussing individual approaches with healthcare professionals remains the most evidence-based path forward. The NAD World concept offers a compelling framework for understanding aging—but translating that framework into personal health strategies requires careful consideration of your unique circumstances, existing conditions, and health goals.
Further Reading
Explore more articles related to this topic: