The quest to understand what are senolytics has captured the attention of researchers and health enthusiasts alike. These compounds represent one of the most intriguing frontiers in aging research, offering a new lens through which we can examine the aging process itself. But separating scientific reality from marketing hype requires a clear-eyed look at what we actually know.
This guide breaks down the science of senolytics in accessible terms, explores the role of quercetin and fisetin as potential senolytic agents, and helps you understand where this research stands today.
What Are Senolytics? (Quick Answer)
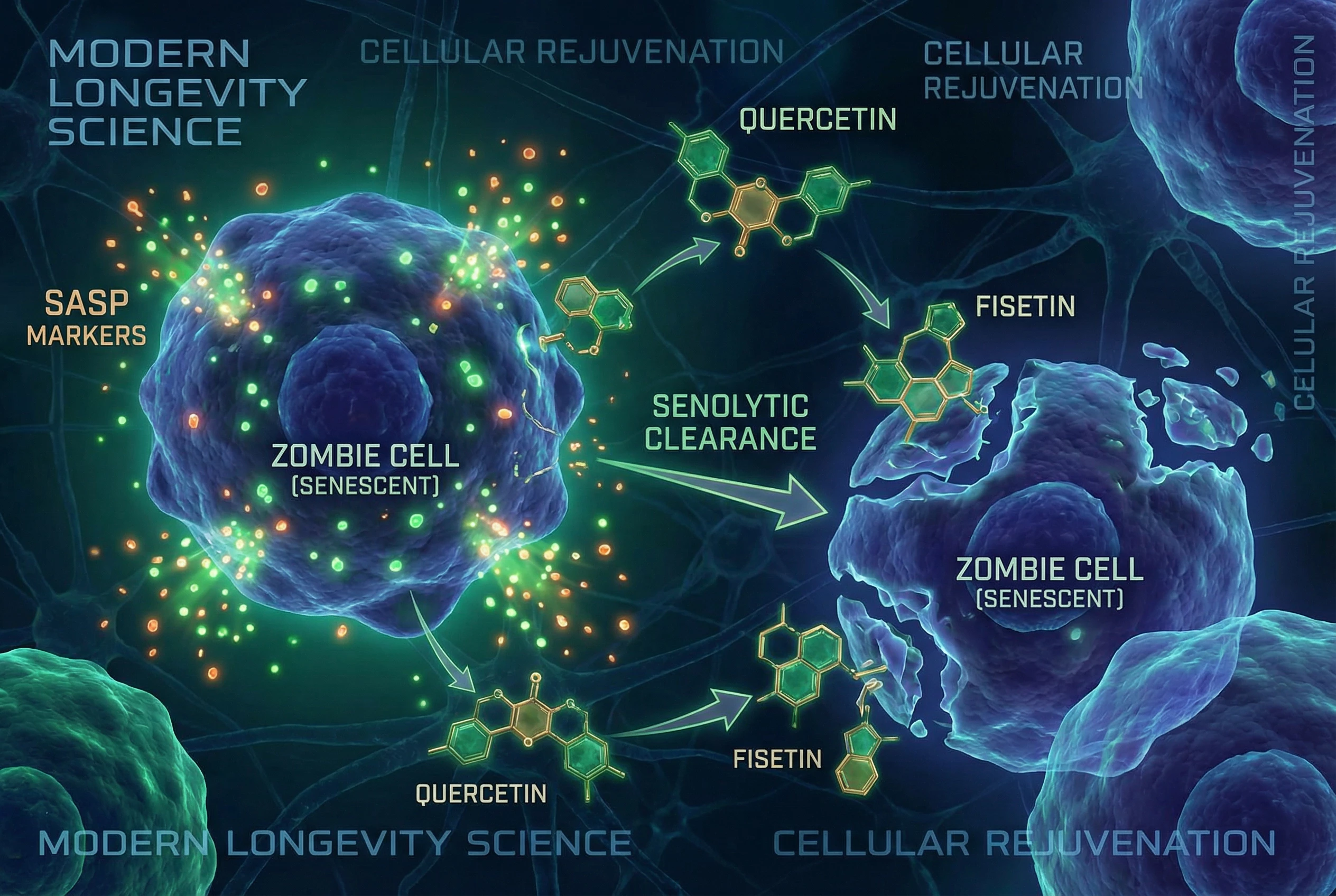
Senescent cells, often called “zombie cells,” are cells that have stopped dividing but do not die. They accumulate in the body, release inflammatory and tissue-degrading molecules, and contribute to age-related health issues.
Senolytics are drugs designed to specifically target and eliminate senescent cells, which have ceased cell division and can contribute to age-related health issues. These damaged cells accumulate with age and secrete inflammatory molecules that can affect surrounding cells and tissues. Researchers have linked senescent cell buildup to various age related diseases, and this accumulation is particularly relevant in older people, contributing to tissue degeneration and age-related diseases, though the relationship is still being mapped out.
The concept is straightforward: if we can selectively clear these dysfunctional cells, we might slow some aspects of the aging process or reduce its detrimental effects. Senolytics are being studied because they target fundamental aging processes that drive cellular senescence and organ decline. This idea has driven significant research investment over the past decade, with promising results in mice and early-stage human studies.
However, it’s crucial to understand that senolytics remain experimental. Most robust data come from animal models, and no senolytic drug has received broad regulatory approval specifically for anti-aging purposes. The compounds showing promise in laboratory settings don’t automatically translate to benefits when consumed as dietary supplements. Early clinical trials have indicated that senolytics can reduce senescent cells in humans and alleviate symptoms related to frailty and age-related conditions.
Quercetin and fisetin are two plant based compounds frequently discussed as “natural” senolytics. These flavonoids appear in many foods and are widely available as supplements. But here’s the catch: the doses used in research studies are often far higher than typical dietary intake, and direct human evidence for senescent cell clearance with these compounds is limited.
Key Takeaway: Senolytics are experimental tools being studied to help clear zombie cells. They are not yet proven anti-aging cures, and most evidence comes from cell cultures and animal studies rather than large human trials.
What Are Zombie (Senescent) Cells and Why Do They Matter?
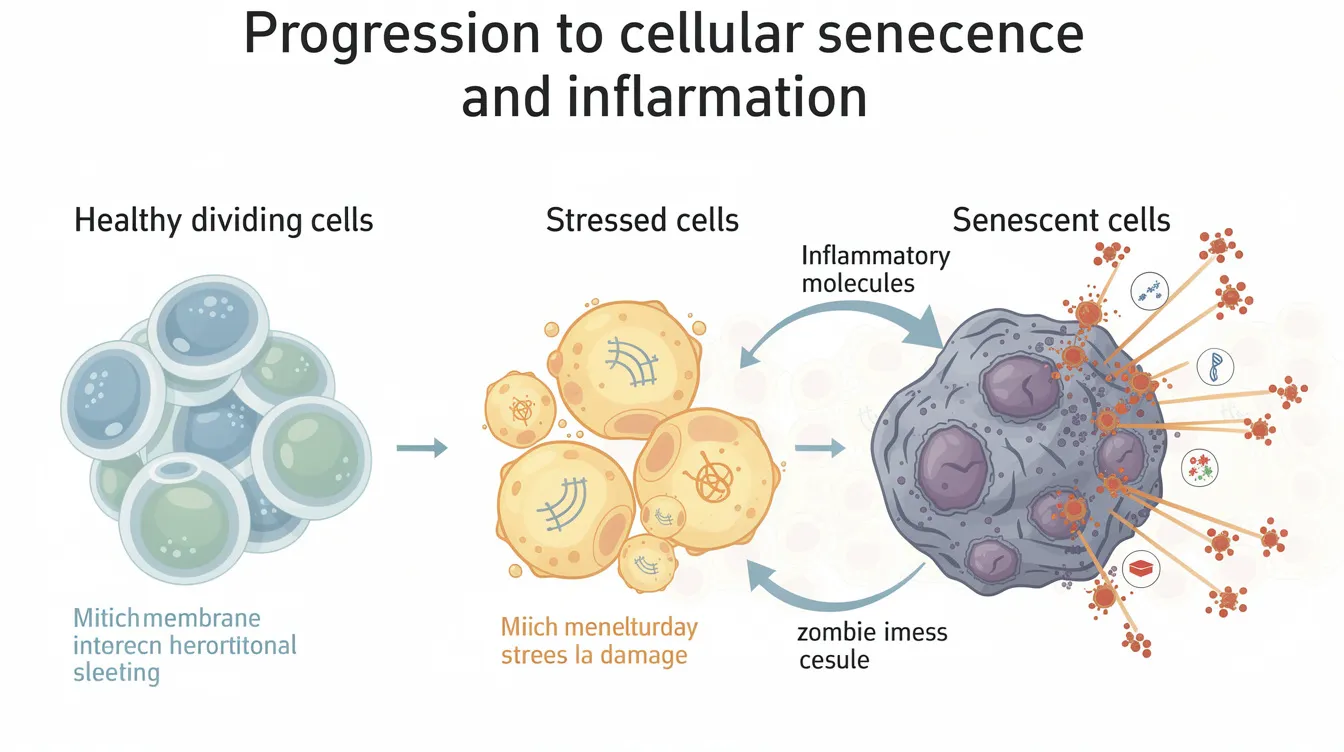
Cellular senescence is a state where cells stop dividing in response to various stressors but remain metabolically active. Unlike apoptosis, where damaged cells are cleared away, senescent cells persist in tissues—alive but no longer contributing to normal tissue repair or function. This is where the “zombie” nickname comes from: they’re not fully dead, but they’re not living normally either.
These cells don’t just sit quietly. They actively influence their environment by secreting a complex mixture of molecules that affects neighboring tissues, immune health, and biological processes throughout the body.
Common triggers of senescence include:
Telomere attrition (chromosomes shortening with each cell division until they hit a critical threshold)
Oxidative stress from accumulated dna damage
Oncogene activation (genes that could drive cancer if the cell kept dividing)
Chemotherapy or radiation exposure
Chronic inflammation and metabolic dysfunction
The Senescence-Associated Secretory Phenotype (SASP)
What makes senescent cells particularly problematic is their secretory behavior. The SASP includes:
| SASP Component | Examples | Effects |
|---|---|---|
| Pro-inflammatory cytokines | IL-6, IL-8, IL-1 | Drive chronic inflammation |
| Chemokines | Various signaling molecules | Recruit immune cells |
| Growth factors | Multiple types | Alter tissue remodeling |
| Proteases | Matrix metalloproteinases | Degrade surrounding tissue |
| This inflammatory cocktail contributes to what researchers call “inflammaging”—the chronic, low-grade inflammation associated with aging. Over time, these secreted factors can promote mitochondrial dysfunction, impair tissue repair, and create an environment conducive to age related conditions. |
The Double-Edged Nature of Senescence
Here’s where it gets complicated: senescence isn’t entirely bad. In the short term, it serves important protective functions. Senescent cells help suppress cancer by forcing potentially dangerous cells to stop dividing. They also contribute to wound healing and proper tissue repair during acute injury.
The problem emerges when senescent cells accumulate over decades. What starts as a protective brake becomes a source of chronic dysfunction. Research has associated elevated senescent cell burden with osteoarthritis, arthritis, atherosclerosis, fibrosis, brain aging, and other conditions—though establishing direct causation in humans remains an active area of investigation. The accumulation of senescent cells is linked to various age-related diseases, including cancer, diabetes, and frailty.
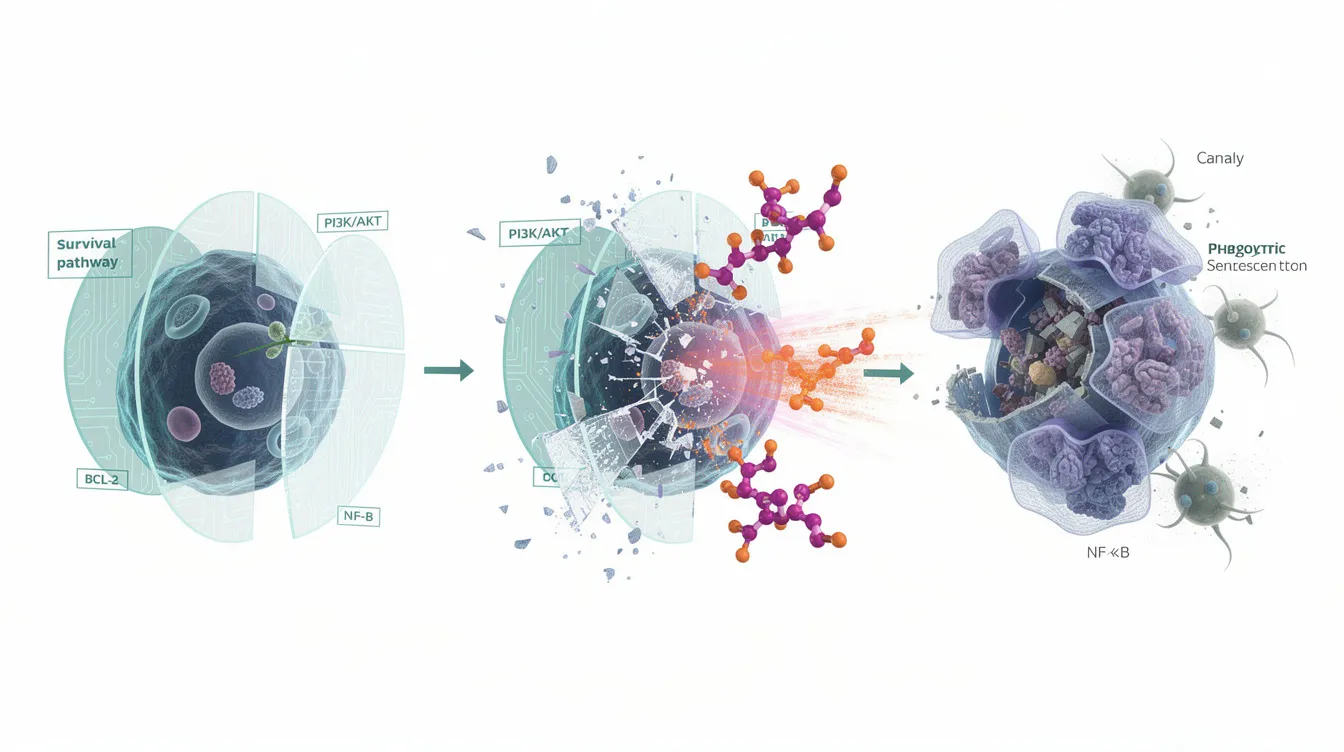
How Senolytics Work: Targeting Senescent Cells
Normal cells have built-in mechanisms for programmed cell death called apoptosis. When cells become damaged beyond repair, they typically self-destruct for the good of the organism. Senescent cells, however, have learned to resist this fate.
These aging cell populations upregulate what researchers call “Senescent Cell Anti-Apoptotic Pathways” or SCAPs. Think of it as senescent cells developing shields against the body’s normal cleanup mechanisms. Senolytics work by disabling these shields, allowing the natural death process to proceed.
Major Senolytic Strategies
Researchers have identified several approaches to targeting senescent cells:
BCL-2/BCL-XL Inhibition: These proteins act as gatekeepers preventing apoptosis. By blocking them, senolytics can remove this protection and allow senescent cells to die.
Tyrosine Kinase Inhibition: Certain kinases transmit survival signals in senescent cells. Blocking these pathways (as dasatinib does) disrupts the cell’s ability to maintain its survival state.
FOXO4-p53 Pathway Modulation: In senescent cells, FOXO4 binds to p53 and prevents it from triggering cell death. Disrupting this interaction frees p53 to activate apoptotic programs.
Metabolic Targeting: Senescent cells have distinct metabolic profiles that can potentially be exploited for selective targeting.
The Dasatinib + Quercetin Combination
The most-studied senolytic combination pairs dasatinib (a tyrosine kinase inhibitor originally approved for certain leukemias) with quercetin (a dietary flavonoid). Mayo clinic researchers pioneered this approach in landmark mouse studies, finding that intermittent treatment could improve markers of healthy aging.
The theory is that dasatinib hits certain survival pathways while quercetin affects others, creating a broader attack on the senescent cell’s defenses. This “hit-and-run” approach—treatment for a few days followed by extended breaks—emerged from observations that senolytic effects persisted after the compounds were cleared from the body.
Senolytics vs. Senomorphics
It’s worth distinguishing between senolytics, which eliminate senescent cells entirely, and senomorphics, which modulate the SASP without killing the cells. Compounds like rapamycin and metformin are sometimes discussed as having senomorphic-like properties—they may reduce inflammatory signaling from senescent cells rather than eliminating them. Both approaches have potential merit, and future therapies might combine them.
Key Senolytic Compounds Under Study
Most robust senolytic findings come from animal and cell culture studies, with human clinical trials still in early phases. Understanding which compounds are being studied—and at what stage—helps calibrate expectations about this field.
Dasatinib
Originally FDA-approved for chronic myeloid leukemia and Philadelphia chromosome-positive acute lymphoblastic leukemia, dasatinib was repurposed as a senolytic after screening studies identified its activity against senescent cells. The landmark Mayo Clinic studies from 2015 onward showed that dasatinib combined with quercetin extended both median lifespan and healthspan in aged mice when administered intermittently.
Small pilot studies in humans with idiopathic pulmonary fibrosis have shown short-term improvements in physical function, including 6-minute walk distance. Additional trials in diabetic kidney disease have reported reduced senescence markers in tissue biopsies. However, these remain small, open label studies—not the large controlled trials needed for definitive conclusions.
Quercetin
This dietary flavonoid occurs naturally in onions, apples, capers, kale, and various berries. Beyond its role in senolytic combinations, quercetin has been studied for decades as an antioxidant and anti-inflammatory compound. Cell culture studies show that quercetin can reduce markers of senescence at high doses, though the concentrations used often exceed what typical supplementation achieves in human blood.
The challenge with quercetin lies in bioavailability—how much of the ingested compound actually reaches tissues at active concentrations. Standard quercetin supplement doses yield blood levels substantially lower than those used in cell culture experiments, raising questions about real-world efficacy. Many over-the-counter quercetin supplements are sold at lower doses than those used in clinical studies, but advanced formulations may allow for effective lower doses.
Quercetin may be better for heart health, while fisetin may be better for general healthy aging support, although there is some overlap.
Fisetin
Found primarily in strawberries, apples, persimmons, and cucumbers, fisetin emerged as a promising senolytic candidate in late 2010s research. Studies in obese mice and aged mice showed reduced senescent cell burden following fisetin treatment. Some mouse models demonstrated extended remaining lifespan.
Multiple human trials investigating fisetin are registered and ongoing as of 2026, though definitive clinical data haven’t yet been published. Fisetin appears to have relatively low toxicity in preclinical studies, making it an attractive candidate for further research.
Navitoclax (ABT-263)
This BCL-2/BCL-XL inhibitor shows strong senolytic activity in preclinical mouse models but carries significant toxicity concerns. Because BCL-XL is also essential for platelet survival, navitoclax can cause thrombocytopenia (low platelet counts) and related hematological problems. These dose-limiting toxicities restrict its potential for routine clinical use, though it remains valuable as a research tool.
Emerging Precision Senolytics
Next-generation approaches aim for tissue-specific targeting. UBX1325, for example, is being developed for retinal diseases including diabetic macular edema and age-related macular degeneration. Early trials focus on safety and visual acuity endpoints, with preliminary data showing promise—though long-term safety remains unknown.
These precision senolytics represent a strategic shift toward localized treatment that maximizes efficacy in specific organs while minimizing systemic exposure.
Quercetin and Fisetin: Natural Flavonoids with Senolytic Potential
Quercetin and fisetin belong to the flavonoid family of plant compounds, characterized by their polyphenolic structure and broad biological activity. Both appear naturally in the diet and have been formulated into dietary supplements marketed for various health benefits.
Food Sources
| Compound | Rich Food Sources |
|---|---|
| Quercetin | Onions, apples, capers, kale, berries, broccoli |
| Fisetin | Strawberries, apples, persimmons, cucumbers, grapes |
| The amounts found in typical servings are much lower than doses used in senolytic research—you’d need to eat impractical quantities of strawberries to match the fisetin doses given to mice in lifespan studies. |
General Biochemical Properties
Both compounds demonstrate:
Antioxidant activity: Scavenging reactive oxygen species that contribute to cellular damage
Anti-inflammatory effects: Modulating pathways including NF-κB that drive inflammation
Enzyme modulation: Affecting various enzymes involved in metabolism and cellular signaling
These properties are well-documented independent of any senolytic effects. The challenge for researchers is distinguishing whether observed health benefits come from general antioxidant and anti-inflammatory activity or from specific senolytic action.
Preclinical Evidence
Cell culture studies show that quercetin and fisetin, at concentrations typically in the 10-100 μM range, can selectively impact senescent cells under controlled conditions. Researchers found reduced senescence markers and increased senescent cell death in various cell types.
Animal studies extend these findings. In one notable study, fisetin treatment in aged mice reduced senescent cell frequency across multiple tissues. The quercetin supplement combined with dasatinib has shown effects on senescent cell burden in mouse adipose tissue, with some studies reporting 30-70% reductions in senescence markers.
The Translation Gap
Here’s where caution is warranted. Peak plasma concentrations of quercetin from standard supplement doses typically reach only 0.5-2 μM—far below the 10-100 μM concentrations showing clear senolytic effects in lab dishes. Whether supplement doses actually achieve senolytic tissue concentrations in humans remains an open question.
The supplement industry has enthusiastically adopted quercetin and fisetin as “natural senolytics,” but this extrapolates from preclinical findings to human anti-aging effects without the connecting evidence. Controlled human data directly demonstrating senescent cell clearance with standalone quercetin or fisetin are limited as of 2026.

What Does the Research Say About Senolytics and Health?
Interpreting senolytic research requires understanding the evidence hierarchy. The most convincing outcomes come from carefully controlled animal studies—primarily in mice—followed by smaller, preliminary human trials. Extrapolating from mouse aging studies to human health is complex because rodent biology doesn’t perfectly mirror human physiology.
Preclinical Findings by System
Metabolic Health
Senescent adipocytes contribute significantly to adipose tissue inflammation. Mouse studies show that senolytic treatment can improve insulin sensitivity, reduce adipose inflammation, and improve glucose handling. Whether this translates to meaningful metabolic improvements in humans requires larger studies.
Cardiovascular Function
Researchers found that clearing senescent endothelial cells and vascular smooth muscle cells improved vascular function in animal models. Reduced atherosclerotic burden and improved endothelial function have been reported, though no large human cardiovascular outcome trials exist.
Musculoskeletal Health
Senescent cells in muscle tissue appear to impair regeneration and contribute to sarcopenia. Senolytic treatment in mice has shown improvements in physical function, muscle strength, and in some studies, muscle mass. Bone resorption and remodeling are also affected by senescent osteoblasts and osteoclasts.
Brain Function
Neuroinflammation driven by senescent glial cells is implicated in cognitive aging and neurodegenerative diseases. Mouse models of Alzheimer’s disease showed some improvements in pathology and brain aging markers following senolytic treatment. However, getting senolytics across the blood-brain barrier in adequate concentrations remains challenging.
Pulmonary Disease
The most clinically advanced senolytic work involves idiopathic pulmonary fibrosis. A pilot study of dasatinib + quercetin in IPF patients showed short-term improvements in 6-minute walk distance and reduced fibrosis markers. This represents the most robust human senolytic data available, though it was small and uncontrolled.
Human Clinical Data: Current Status
| Indication | Compound(s) | Trial Phase | Key Findings |
|---|---|---|---|
| Idiopathic Pulmonary Fibrosis | D+Q | Phase 1/2 | Improved walk distance, reduced senescence markers |
| Diabetic Kidney Disease | D+Q | Phase 1/2 | Reduced adipose tissue senescence markers |
| Age-Related Macular Degeneration | UBX1325 | Phase 1/2 | Safety established, visual acuity endpoints studied |
| Frailty in Older Adults | Fisetin | Phase 2 | Ongoing, results pending |
Critical Uncertainties
Significant questions remain unanswered even for the most-studied senolytics:
What is the optimal age to initiate senolytic treatment?
Should treatment be intermittent or continuous?
What doses balance efficacy against potential toxicity in humans?
How do responses vary by sex, genetics, and underlying health condition?
What are the long-term consequences of reducing protective senescent cells?
Regulatory agencies have not approved any senolytic broadly for anti-aging indications. Current trials focus on specific diseases rather than general longevity or healthy aging claims.
Risks, Unknowns, and Safety Considerations
Clearing senescent cells is biologically powerful. The same mechanisms that make senolytics potentially beneficial also mean they could carry trade-offs, particularly when used without medical supervision.
Protective Roles That Could Be Compromised
Senescence serves important functions that indiscriminate elimination might disrupt:
Tumor suppression: Senescence forces potentially cancerous cells to stop dividing. Clearing these cells theoretically could allow some pre-cancerous cells to escape this protective brake.
Wound healing: Senescent cells contribute to proper tissue repair and scar formation during acute injury.
Tissue remodeling: Short-term senescence helps resolve fibrosis in some contexts and coordinates developmental processes.
Known and Plausible Side Effects
From prescription senolytics: Navitoclax and similar BCL-2 inhibitors cause cytopenias—particularly thrombocytopenia. Dasatinib can affect bone remodeling and has known off-target effects on non-senescent cells expressing its target kinases.
From high doses of flavonoid supplements: Drug interactions are possible. Quercetin inhibits P-glycoprotein, potentially affecting medications that depend on this transporter. Both quercetin and dasatinib are metabolized hepatically and could interact with cytochrome P450 enzyme substrates.
Supplement-Specific Concerns
Over-the-counter senolytic supplements present particular challenges:
Standardization and purity are not guaranteed
Dosing recommendations lack rigorous evidence bases
Marketing claims often dramatically exceed scientific support
Formulations may not match those used in controlled research
Individual responses vary considerably based on genetics and absorption
Individual Variability
Emerging research suggests that age, sex, underlying conditions, and genetic background all influence senolytic response. What works in a healthy older person might have different effects in someone with diabetes or taking immunosuppressive medications. A one-size-fits-all approach is not supported by current science.
Anyone considering high-dose flavonoid supplements or experimental senolytic approaches should consult a qualified healthcare professional, particularly those who take prescription medications, have chronic illness, cancer history, or are pregnant, breastfeeding, or immunocompromised.
Evidence-Based Lifestyle Strategies That Influence Senescence
While senolytic drugs remain experimental, several well-supported lifestyle habits influence cellular health, inflammation, and metabolic function with much broader evidence and established safety profiles.
Physical Activity
Regular exercise may be the closest thing to a “natural senolytic” intervention. Research links physical activity to:
Reduced inflammatory markers and improved immune system function
Enhanced mitochondrial function and energy production
Lower senescent cell burden in muscle and other tissues (in animal studies)
Improved metabolic health and insulin sensitivity
20-30% reductions in all-cause mortality in large prospective studies
These benefits operate through multiple pathways, likely including but not limited to effects on senescence. Even if the senescence-specific effects are modest, the overall health benefits are well-established.
Dietary Patterns
Mediterranean-style diets rich in diverse plant foods provide natural flavonoids including quercetin and fisetin at food-level doses. More importantly, these dietary patterns deliver:
Fiber and polyphenols with anti-inflammatory effects
Healthy fats that support healthy aging and cardiovascular function
A synergistic combination likely exceeding what any single isolated compound provides
Major studies associate Mediterranean diet adherence with 20-30% reductions in all-cause mortality and lower rates of cardiovascular disease, diabetes, and cognitive decline.
Sleep Quality
Chronic sleep deprivation associates with increased oxidative stress, impaired immune function, elevated inflammatory markers, and accelerated biological aging. Targeting 7-9 hours of consistent sleep supports cellular repair processes that occur during rest. Even single nights of poor sleep measurably increase stress markers.
Stress Management and Social Connection
Chronic psychological stress elevates cortisol, increases oxidative stress, and drives systemic inflammation. Social isolation carries mortality risks comparable to smoking or obesity. While the mechanisms linking stress to cellular senescence are still being explored, the broader effects on health and longevity are indisputable.

How to Think About Senolytic Supplements as a Beginner
Interest in senolytics comes from a genuine desire to support healthy aging and maintain function—legitimate and understandable goals. However, this motivation can make it easier to accept claims with insufficient evidence, particularly when framed in scientific language.
Questions to Ask Before Considering Any Senolytic Supplement
What human data exist for this exact product at this exact dose?
Are claims based on cell or mouse studies that haven’t been replicated in people?
Are there known interactions with medications I currently take?
Has a qualified healthcare professional reviewed the evidence?
Who funded the research being cited, and are there conflicts of interest?
What is the regulatory status of the product?
A Practical Hierarchy of Intervention
Before layering experimental approaches, consider this sequence:
Optimize foundational behaviors: Sleep, physical activity, dietary pattern, stress management, social connection
Address existing medical conditions with established treatments
Only then consider adding experimental interventions like senolytics
Clinical Trials as an Alternative
Participating in well-designed clinical trials at academic research centers may be a safer way to access senolytic research than self-experimentation with supplements. Trials have institutional review board oversight, informed consent processes, and built-in monitoring for adverse effects. While inclusion criteria may exclude some people and access is geographically limited, trial participation provides access to actual research compounds at studied doses.
Realistic Expectations
Even if senolytics eventually prove beneficial in rigorous trials, they’re unlikely to be magic bullets that dramatically reverse aging. The aging process involves numerous cellular and systemic changes beyond senescent cell accumulation. Senolytics would most likely complement rather than replace lifestyle optimization and standard medicine.
Beginner’s Checklist:
Consult a qualified healthcare professional
Review evidence sources (peer-reviewed studies, not marketing)
Consider optimizing lifestyle factors first
Avoid megadoses without medical supervision
Monitor for adverse effects and report them
Keep expectations realistic
Open Questions and Future Directions in Senolytic Research
Senolytic research is advancing rapidly, supported by major initiatives including the NIH-funded Cellular Senescence Network (SenNet), which is systematically mapping senescent cells across human tissues. International consortia and leading research centers continue pushing the field forward.
High-Priority Research Questions
How can we selectively target harmful senescent cells while preserving beneficial ones?
Which biomarkers best track senescent burden and therapy response in humans?
What timing (age, disease stage) and dosing schedule is optimal?
How do sex, genetics, and environment affect individual outcomes?
What are the long-term consequences of reduced senescent cell populations?
Emerging Technologies
Researchers are developing safer, more selective delivery systems:
Nanoparticles engineered to accumulate in specific tissues
Antibody-drug conjugates targeting senescent cell surface markers
Approaches exploiting tissue-specific vulnerabilities
These innovations aim to localize senolytics to target organs while minimizing systemic exposure and off-target effects.
Combination Approaches
Rather than senolytics alone, the field increasingly explores combinations with senomorphics, immunotherapies, and regenerative medicine. Complex age related diseases involve multiple pathways; targeting cellular senescence alone may be insufficient. However, combining powerful biological interventions also increases the risk of unforeseen interactions.
The Path Forward
Quercetin, fisetin, and other senolytics have opened a new window into aging biology. The fundamental mechanisms of senescence are now better understood than ever, and the tools to intervene continue to improve. But responsible use requires waiting for more rigorous, long-term human data.
The field needs large prospective trials measuring hard outcomes—disease incidence, lifespan, quality of life—rather than surrogate markers alone. Such trials require years to complete but are necessary to establish clinical benefit and safety for broader populations.
Stay curious. Follow reputable sources including peer-reviewed journals and major medical centers like the Mayo Clinic and Buck Institute. View senolytics as a fascinating, evolving field of research rather than a finished therapeutic toolkit.
References and Suggested Further Reading
Understanding senolytics requires engaging with primary scientific literature. The following categories of sources provide reliable information:
Landmark Studies and Reviews
Research from Mayo Clinic (senior author James Kirkland et al.) on dasatinib + quercetin represents foundational work in the field. Key publications appeared in Nature Medicine and Aging Cell from 2015 onward. These studies established proof-of-concept that senolytic treatment could improve healthspan in mice.
Fisetin research, including work published in EBioMedicine (2018) and subsequent papers, documented effects on senescent cell burden and remaining lifespan in aged mice.
Clinical Trial Reports
The first in human senolytic trials in idiopathic pulmonary fibrosis (published in Lancet-affiliated journals and clinical practice publications) provide crucial early data on safety and preliminary efficacy signals.
National Institutes of Health clinical trial registries (ClinicalTrials.gov) list ongoing and completed senolytic trials with detailed protocols and, when available, results.
Accessible Overview Resources
The Buck Institute for Research on Aging and Mayo Clinic’s research blogs offer accessible summaries for non-specialists. Review articles in journals like Nature Reviews Drug Discovery and Cell Metabolism provide comprehensive overviews for readers comfortable with scientific terminology.
Evidence Quality Indicators
When reviewing sources, note whether findings come from:
Cell culture studies (most preliminary)
Mouse models (informative but not directly applicable to humans)
Human pilot studies (small sample sizes, often open label)
Randomized controlled trials (strongest human evidence)
This hierarchy helps calibrate confidence in any given claim about senolytics and their potential benefits for human longevity and health.
Further Reading
Explore more articles related to this topic:
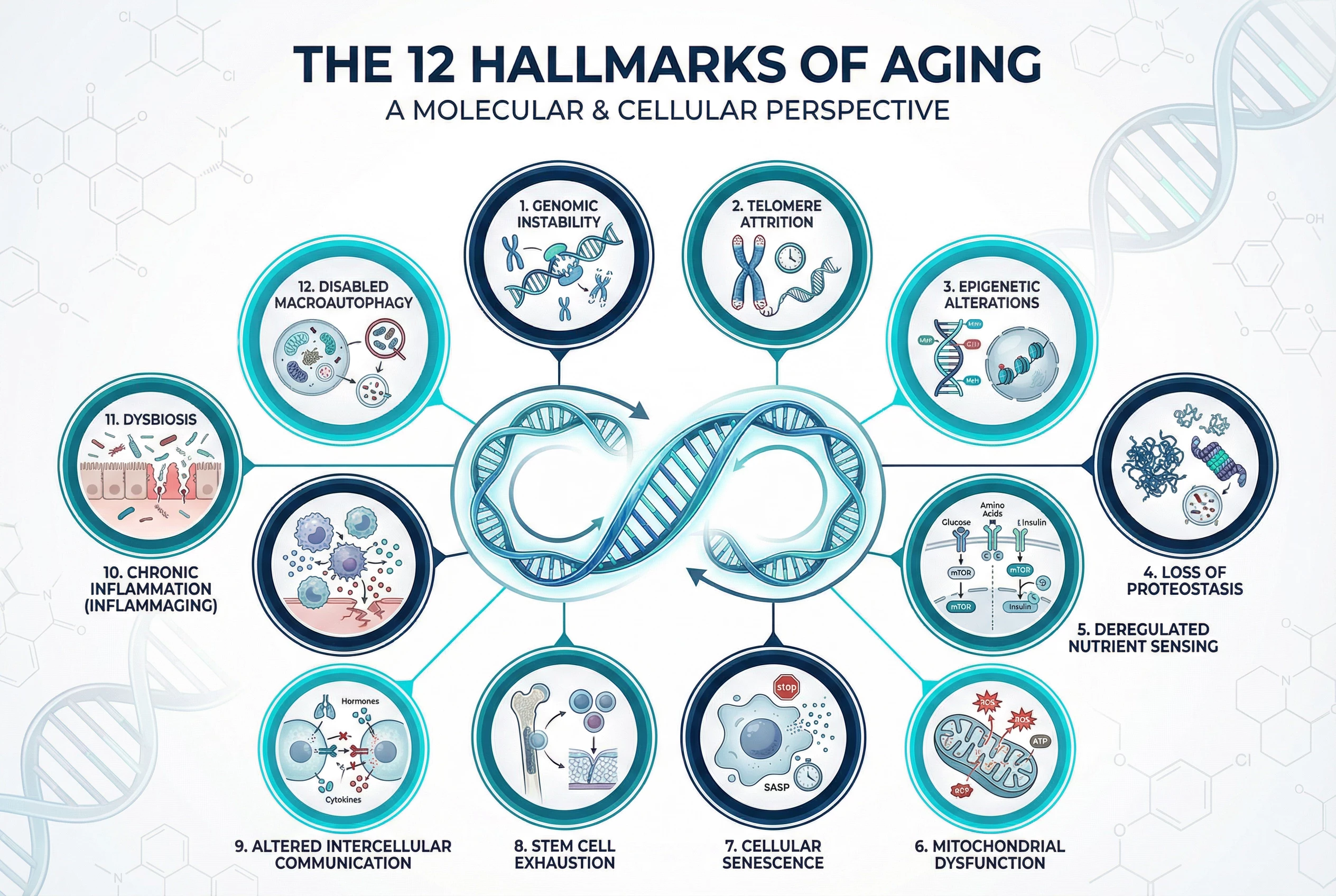
- The 12 Hallmarks of Aging Explained: A Beginner’s Guide to Longevity Science
- The Link Between NMN and Telomere Length: Can You Actually Reverse Biological Age?
- NMN and Urolithin A: The New Frontier of Mitochondrial Health in 2026
- Best Longevity Supplements in 2026: Science-Backed Picks Only
- NMN and Apigenin: The Science of CD38 Inhibition