Introduction: Why the 12 Hallmarks of Aging Matter
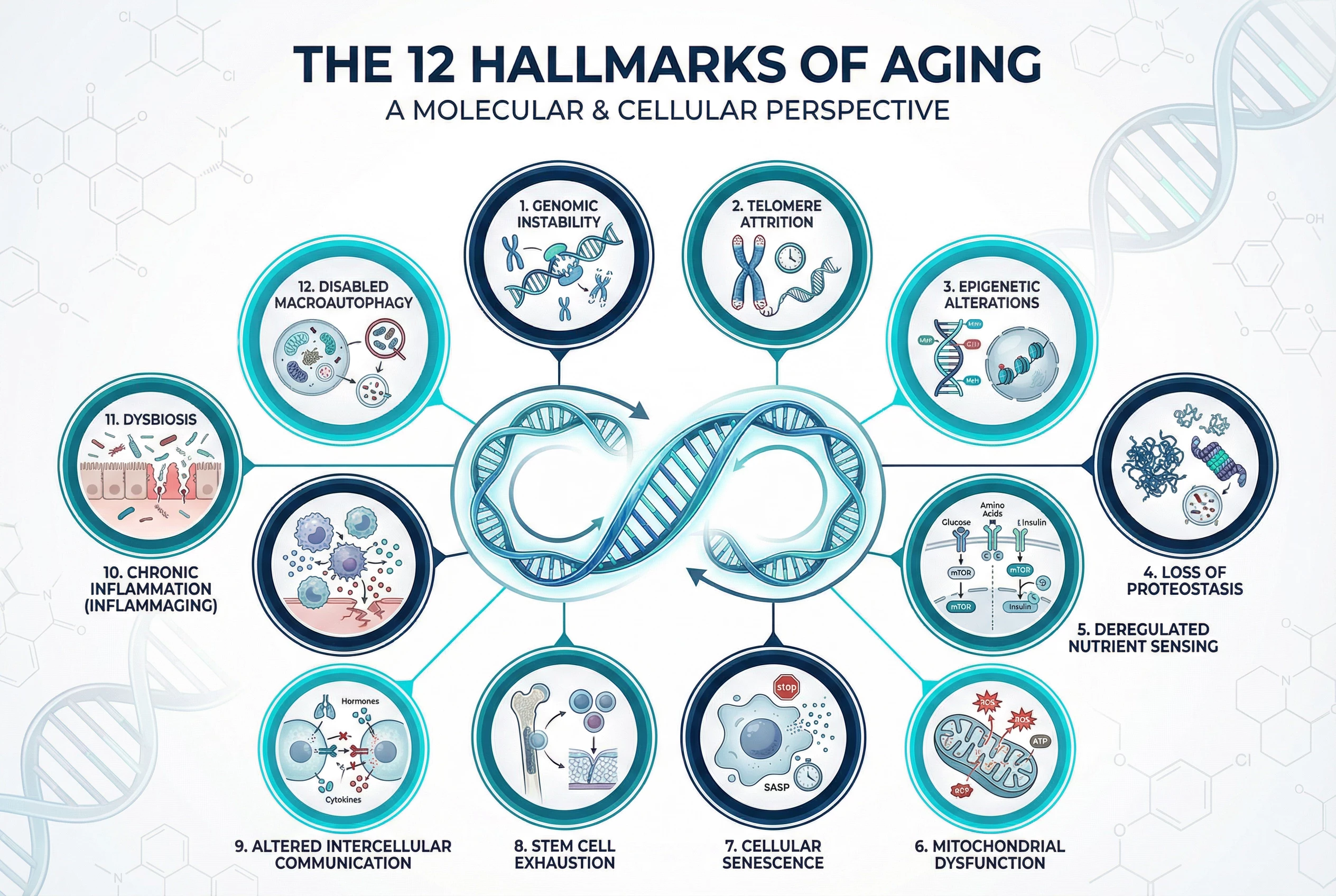
Aging isn’t simply random wear and tear. Decades of research across species—from yeast and worms to mice and humans—have revealed that the aging process follows recurring biological patterns. Scientists call these patterns the 12 hallmarks of aging, a framework that helps explain why our bodies change over time and what drives age related diseases. The 12 hallmarks of aging include genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis.
As people age, they experience increased health problems, including a higher prevalence of illnesses and disabilities. These health problems not only affect individuals but also have a significant impact on society. Older people generally incur more health-related costs than younger people in the workplace and have different requirements from society and government. The proportion of older people in the population is rising globally, with estimates that by 2050, approximately 22% of the world’s population will be aged 60 and older. Population ageing is driven by three main causes: migration, longer life expectancy, and decreased birth rate. This demographic shift poses social and economic challenges, but with the right set of policies, these challenges can be addressed.
The term was popularized in a landmark 2013 paper by López-Otín and colleagues, published in the journal Cell. That original work identified nine hallmarks. By 2023, the framework had expanded to 12, reflecting new discoveries about how the human body ages at the molecular level. The 2013 paper alone has been cited over 11,000 times, underscoring its influence on aging research worldwide.
Important note: This article is educational, not medical advice. Before changing medications, supplements, or treatment approaches, consult with qualified healthcare professionals.
Here are the 12 hallmarks of aging at a glance:
Genomic instability
Telomere attrition
Epigenetic alterations
Loss of proteostasis
Deregulated nutrient sensing
Mitochondrial dysfunction
Cellular senescence
Stem cell exhaustion
Altered intercellular communication
Disabled macroautophagy
Chronic inflammation (inflammaging)
Microbiome dysbiosis
Each hallmark represents a process that changes with age, contributes to functional decline, and—when addressed—may delay features of human ageing. Throughout this guide, we’ll explore each one, discuss practical lifestyle angles where evidence suggests benefits (sleep, physical activity, nutrition, social ties), and highlight open questions in current research.

How Scientists Define a “Hallmark” of Aging
Not every biological change qualifies as a hallmark. To earn that designation, a process must meet three criteria:
It changes with age in observable, measurable ways
Accelerating it worsens aging—speeding up decline or disease
Improving it delays features of aging—extending health span or life span
Consider telomere attrition as an example. Telomeres shorten with each cell division. Research shows that artificially shortening telomeres accelerates cellular aging, while preserving telomere length in some animal models improves health outcomes.
It’s also essential to distinguish between causes of death—heart attacks, stroke, cancer, dementia—and the upstream aging mechanisms that increase disease risk. Genomic instability or cellular senescence aren’t diseases themselves, but they create conditions where diseases become more likely.
These hallmarks don’t operate in isolation. They form feedback loops: mitochondrial dysfunction increases reactive oxygen species production, which contributes to genomic instability, which can trigger cellular senescence. Understanding these connections helps explain why addressing aging requires a systems approach rather than single-target interventions.
Hallmark 1: Genomic Instability (DNA Damage Over a Lifetime)
Every day, your genetic material faces assault. DNA damage accumulates from internal sources—normal metabolism, replication errors—and external sources like UV light, tobacco smoke, ionizing radiation, and environmental toxins. The result is genomic instability, a gradual accumulation of mutations and chromosomal abnormalities.
Your cells have sophisticated DNA repair systems: nucleotide excision repair, base excision repair, and double-strand break repair mechanisms. But repair efficiency may decline with age, allowing more damage to persist.
Human conditions illustrate what happens when DNA repair fails. Xeroderma pigmentosum involves defective repair of UV damage and leads to accelerated aging features. Werner syndrome, caused by mutations in a DNA helicase gene, produces premature aging characteristics in adults. Genomic instability can affect multiple physiological systems, leading to changes in organ function and increased disease risk.
How genomic instability contributes to aging:
Mutations in stem cells reduce renewal capacity
Accumulated damage in somatic cells impairs organ function
Increased cancer risk as cells divide with uncorrected errors
Compromised tissue maintenance over time
Practical considerations:
Research on compounds affecting DNA repair pathways continues in animal models and cell cultures, but these remain experimental. What you can do now: avoid tobacco, limit unnecessary radiation exposure, use sun protection, and follow public health guidelines on environmental toxin exposure.

Hallmark 2: Telomere Attrition (The Shortening “Caps” on Chromosomes)
Telomeres are protective DNA-protein caps at chromosome ends—think of the plastic tips on shoelaces that prevent fraying. Each time cells divide, telomeres naturally shorten due to the “end-replication problem.” Eventually, they become critically short, triggering cells to enter senescence or undergo programmed cell death.
An enzyme called telomerase can extend telomeres. It’s active in germ cells, some stem cells, and many cancer cells. However, most human somatic cells have low telomerase activity, creating a fundamental limit on how often differentiated cells can divide before they stop dividing entirely.
Observational studies suggest that shorter leukocyte telomere length is associated with higher risk of cardiovascular disease, certain cancers, and neurodegeneration. But correlation doesn’t prove causation—telomere length alone doesn’t reliably predict individual lifespan.
Animal research reveals a complex trade-off: artificially increasing telomerase can extend lifespan in some models but may also raise cancer risk, since cancer cells express telomerase to achieve unlimited replication.
Factors associated with telomere shortening:
Chronic psychological stress
Smoking
Physical inactivity
Poor sleep quality
Factors associated with more favorable telomere profiles:
Regular physical activity
Adequate sleep
Social support and connection
Consumer telomere-testing kits exist but cannot reliably predict biological age or future disease risk. Interpret results cautiously and discuss with healthcare professionals.
Hallmark 3: Epigenetic Alterations (Turning Genes On and Off)
Epigenetics refers to chemical tags and structural changes—DNA methylation, histone modification, chromatin remodeling—that influence gene activity without altering the underlying DNA sequence. Think of these as dimmer switches controlling whether genes are expressed strongly, weakly, or not at all.
With age, epigenetic patterns drift. Some genes that should be active become silenced; others that should be quiet become overexpressed. This epigenetic drift contributes to declining cell function and tissue health.
Epigenetic aging clocks represent a major research tool. The Horvath clock, published in 2013, uses DNA methylation patterns at specific genomic sites to estimate biological age. Studies suggest these clock measurements correlate with mortality risk and disease incidence.
However, epigenetic clocks aren’t crystal balls. Different clocks sometimes disagree on estimates, and interventions that reliably and safely reverse clock age in humans remain under investigation.
What research shows:
Diet, smoking, physical activity, and pollution exposure can shift epigenetic patterns
Lifestyle may influence epigenetic aging, though mechanisms are still being clarified
“Younger” epigenetic age isn’t always straightforwardly beneficial—rapid changes can sometimes reflect stress
Practical focus: Rather than chasing specific clock metrics, emphasize robust behaviors associated with good health outcomes in large studies: adequate sleep, regular movement, whole-food eating patterns, social connection, and toxin avoidance.
Hallmark 4: Loss of Proteostasis (Protein Quality Control Breaking Down)
Proteostasis encompasses the cellular systems ensuring proteins are correctly folded, repaired, and removed when damaged. Key players include:
Chaperone proteins that assist proper folding
The ubiquitin-proteasome system that tags and degrades damaged proteins
Autophagy-lysosome pathways that clear larger aggregates and organelles
With aging, misfolded proteins accumulate and form aggregates. This is particularly visible in neurodegenerative conditions: Alzheimer’s disease features amyloid-β plaques and tau tangles; Parkinson’s disease involves α-synuclein aggregates.
Animal experiments show that enhancing chaperone activity or proteasome function can extend lifespan, positioning proteostasis as a promising but early-stage therapeutic target.
The sleep connection: The brain’s glymphatic system—which clears metabolic byproducts and potentially aggregated proteins—is most active during sleep. This provides a direct link between getting enough sleep and protein clearance in the brain.
Supporting proteostasis through lifestyle:
Maintain regular sleep schedules
Manage vascular risk factors (blood pressure, cholesterol, glucose)
Stay cognitively and physically engaged
Follow guidance on preventing head injuries
These habits support long-term brain health and may indirectly support protein quality control systems.
Hallmark 5: Deregulated Nutrient Sensing (How Cells “Read” Food and Energy)
Cells possess nutrient-sensing pathways that interpret signals about energy availability and regulate growth, repair, and metabolism. Key pathways include:
Insulin/IGF-1 signaling – responds to food intake
mTOR – promotes growth when nutrients are abundant
AMPK – activated during energy scarcity
Sirtuins – involved in stress response and metabolism
Deregulated nutrient-sensing disrupts metabolic pathways, which can contribute to diseases like diabetes and metabolic failure. Mitochondrial efficiency also declines with age, resulting in increased oxidative stress and reduced energy production.
Research across species shows that dialing down growth-promoting signals can extend lifespan. Caloric restriction—typically 20-40% fewer calories than unrestricted feeding—has extended healthy lifespan in yeast, worms, flies, and mice. Intermittent fasting protocols show similar effects in some animal models.
In humans, the picture is more complex. Extreme or unsupervised restriction carries real risks: nutrient deficiencies, bone loss, hormonal disruption. This is especially concerning for children, pregnant individuals, older adults, and those with existing medical conditions. Aging reduces the effectiveness of the nutrient-sensing network, leading to metabolic issues such as obesity and diabetes, and other factors such as lifestyle choices, medications, and health conditions can also contribute.
Conceptual framework:
| Growth Mode | Repair Mode |
|---|---|
| High nutrient availability | Nutrient scarcity |
| Elevated mTOR activity | Higher AMPK/autophagy activity |
| Cell proliferation emphasis | Cellular maintenance emphasis |
| Human observational findings: Diets rich in minimally processed plant foods, adequate protein, whole grains, and limited ultra-processed foods are consistently associated with better human health and longevity. A healthy diet provides cells with vital sources of energy and keeps them stable and working as they should. Mediterranean-style eating patterns exemplify this approach. | |
| Open questions: Which aspects matter most—total calories, protein timing, food quality, meal timing? How do genetics, sex, and chronological age modify responses? Research continues. |
Hallmark 6: Mitochondrial Dysfunction (Energy Factories Under Stress)
Mitochondria are cellular organelles that generate ATP—the energy currency powering cellular processes. They also play critical roles in signaling, reactive oxygen species production, and cell death pathways.
Over time, mitochondria accumulate damage:
Mutations in mitochondrial DNA (which lacks some protective mechanisms of nuclear DNA)
Impaired dynamics (fusion and fission processes)
Reduced efficiency in energy production
The result: less energy production and more oxidative stress, which can damage lipids, proteins, and DNA throughout the cell.
Animal studies demonstrate that mitochondrial-targeted interventions—engineered antioxidants, manipulation of mitochondrial dynamics—can influence lifespan. Research with mtDNA mutator mice has shown how accumulated mitochondrial mutations accelerate aging features. Translation to human medicine remains early-stage.
What has strong evidence in humans: Regular physical activity is one of the most robust, low-risk ways to improve mitochondrial function and stimulate mitochondrial biogenesis. This finding comes from decades of exercise physiology research.
Supporting mitochondrial health:
Regular aerobic activity
Resistance training
Adequate recovery and sleep
Avoiding severe overtraining
What remains experimental: Some compounds—CoQ10, NAD+ precursors, certain antioxidants—are under active study. Evidence for broad “anti-aging” benefits in healthy humans isn’t yet definitive.

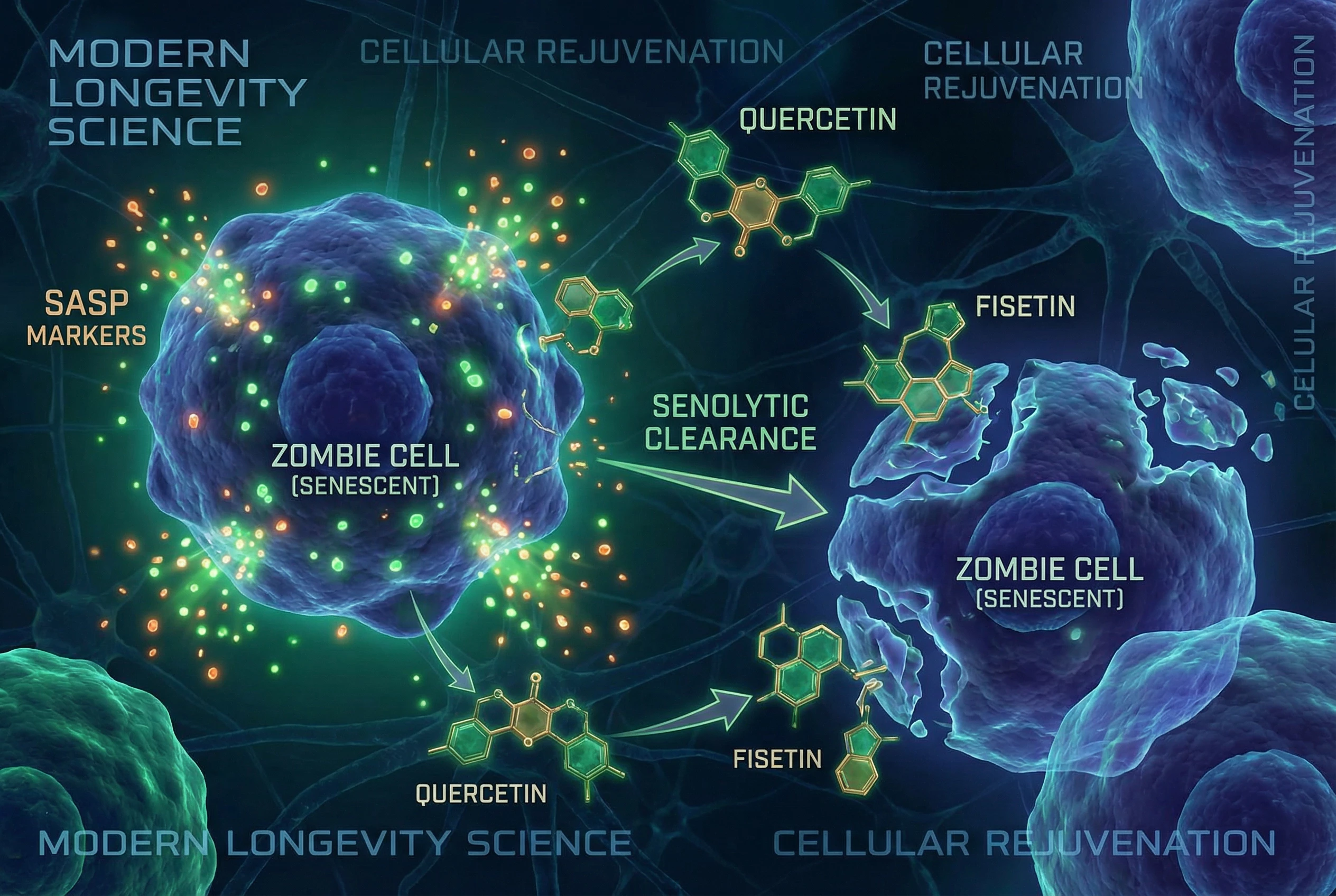
Hallmark 7: Cellular Senescence (Cells That Stop Dividing but Don’t Leave)
Cellular senescence is a state where cells permanently exit the cell cycle—they stop dividing—in response to stressors like DNA damage, telomere shortening, or oncogene activation. Unlike dead cells, senescent cells remain metabolically active.
The problem: senescent cells secrete a mix of inflammatory molecules, growth factors, and enzymes called the senescence-associated secretory phenotype (SASP). This cocktail alters nearby tissues and contributes to chronic inflammation and tissue dysfunction.
Senescence is a double-edged sword. It can prevent cancer by stopping damaged cells from dividing, and it plays beneficial roles in wound healing and embryonic development. Problems emerge when senescent cells accumulate faster than the body can clear them.
Research developments:
Senolytic drugs—compounds that selectively eliminate senescent cells—have extended health span and delayed age related diseases in mouse studies. Multiple early-phase human trials are now underway.
However, current senolytics are experimental with potential side effects. They’re not approved as general longevity treatments and should not be self-administered outside clinical trials.
Lifestyle connections:
Maintaining metabolic health may reduce senescence drivers
Avoiding smoking limits DNA damage that triggers senescence
Regular physical activity supports tissue health
Managing UV exposure reduces skin cell damage
Open questions: How many senescent cells constitute “too many”? Which tissues matter most for health outcomes? What would be the optimal dosing schedule if senolytics prove effective?
Hallmark 8: Stem Cell Exhaustion (Declining Tissue Renewal)
Adult tissues depend on stem and progenitor cells for repair and renewal. Your blood, skin, gut lining, and muscles continuously replace old or damaged cells using these regenerative reservoirs.
With aging, stem cells can decline in number, function, or both. The consequences are visible: slower wound healing, reduced blood-forming capacity, changes in muscle and bone maintenance that many older adults experience.
Specific examples:
Hematopoietic stem cells in bone marrow skew toward certain blood cell types with age, potentially weakening immune responses
Satellite cells in muscle show reduced regenerative ability, contributing to age-related muscle loss and declining grip strength
Animal experiments have demonstrated ways to re-energize stem cells—modifying signaling pathways, parabiosis experiments joining young and old circulations, manipulating systemic factors. Translating these to safe human therapies remains challenging.
A note on stem cell clinics: Some stem cell therapies are legitimate and regulated for specific conditions. Others marketed directly to consumers may be unproven or risky. Verify regulatory approvals and discuss any treatment with qualified clinicians before proceeding.
Supporting tissue health:
Physical activity (especially resistance training)
Adequate protein intake (particularly important for older adults per current nutritional guidelines)
Avoiding smoking
Managing chronic conditions like diabetes and heart disease
These habits may help preserve stem-cell niches and support ongoing tissue renewal.
Hallmark 9: Altered Intercellular Communication (Signals Going Off-Key)
Cells communicate constantly through hormones, neurotransmitters, immune signals (cytokines), and direct cell-to-cell contacts. This intercellular communication coordinates functions across tissues and organs.
Age-related changes disrupt these signals:
Increased pro-inflammatory signaling (“inflammaging”)
Impaired endocrine signaling, including insulin resistance
Altered neural signaling
Changes in extracellular vesicles that transport molecular messages
Familiar examples:
Chronic low-grade inflammation is associated with higher cardiovascular disease risk, frailty, and faster functional decline. Less responsive immune signaling contributes to weaker responses to infections and vaccines in older adults.
The blood and extracellular fluid carry “aging signals” between organs. Parabiosis experiments in mice—where young and old circulations are surgically joined—demonstrate that systemic factors profoundly influence aging across tissues.
Lifestyle connections:
Social isolation and loneliness correlate with higher mortality and elevated inflammation markers
Regular physical activity and a healthy diet associate with more favorable inflammatory profiles
Chronic stress disrupts hormonal signaling and elevates cortisol
Questions worth considering: How might environmental factors—air pollution, work schedules, social connection with loved ones—shape these intercellular signals? Could changing your environment be as important as targeting individual cells?
Hallmark 10: Disabled Macroautophagy (Reduced Cellular “Self-Cleaning”)
Macroautophagy (commonly called autophagy) is the cell’s self-cleaning process. Cells enclose damaged components—misfolded proteins, dysfunctional organelles—in vesicles and deliver them to lysosomes for breakdown and recycling.
Autophagy activity generally declines with age across many tissues. The result: buildup of cellular “junk” and reduced flexibility in responding to stress.
Animal model research shows that enhancing autophagy—through genetic manipulation, fasting, or certain compounds—links to increased lifespan and resistance to age-related conditions.
Connection to nutrient sensing: Autophagy ramps up when nutrient availability drops and mTOR activity decreases. This connects directly to deregulated nutrient sensing and explains some benefits attributed to caloric restriction and fasting protocols.
Human-relevant aspects:
Intermittent fasting and certain dietary patterns can influence autophagy markers, but optimal regimens, long-term safety, and impacts on mortality remain unclear.
Nuanced guidance:
Focus on overall metabolic health
Avoid chronic overnutrition
Consider overnight fasting windows that fit your medical status and lifestyle
Consult healthcare providers if you have diabetes, are pregnant, have history of eating disorders, or take specific medications
Important caveat: More autophagy isn’t always better. Excessive stress and malnutrition can push autophagy to harmful levels—another example of complex, non-linear dose-response relationships in cell biology.
Hallmark 11: Chronic Inflammation (Inflammaging)
“Inflammaging” describes the persistent, low-grade inflammation common in many older adults. Unlike acute inflammation—the body’s healthy response to injury or infection—chronic inflammation simmers continuously, damaging tissues over time.
Major sources:
Senescent cells releasing SASP factors
Misfolded proteins activating immune responses
Changes in gut barrier function
Immune system remodeling with age
Environmental exposures (air pollution, smoking)
What studies show:
Higher levels of inflammatory markers like C-reactive protein (CRP) and interleukin-6 are linked with increased risk of frailty, cardiovascular disease, and mortality. Chronic inflammation has been connected to nearly every major chronic condition—heart disease, arthritis, diabetes, Alzheimer’s.
Anti-inflammatory drugs help specific conditions but aren’t proven as broad anti-aging therapies. Chronic use carries risks: increased bleeding, kidney effects. Medical guidance is essential.
Evidence-based strategies linked with lower inflammation:
Regular physical activity (150+ minutes moderate aerobic activity weekly)
Sufficient sleep (roughly 7-9 hours for most adults)
Smoking avoidance
Maintaining healthy weight (avoiding extra weight that promotes inflammation)
Dietary patterns rich in vegetables, fruits, whole grains, unsaturated fats
Psychosocial dimensions: Chronic stress, loneliness, and adverse childhood experiences associate with elevated inflammation, highlighting how mental health and social connection influence biological aging.
Hallmark 12: Microbiome Dysbiosis (Shifts in Our Inner Ecosystem)
The human microbiome encompasses the microorganisms—bacteria, fungi, viruses—residing in and on your body. The gut microbiome particularly influences digestion, immune system education, and metabolite production affecting overall human health.
Age-related changes:
Reduced microbial diversity in some older populations
Overgrowth of certain bacterial species
Thinning of protective gut mucus layer
Associations with frailty, cognitive changes, and inflammation
Critical caveat: Which microbiome changes cause aging versus result from aging and disease isn’t fully established. Animal interventions like fecal transplants that “rejuvenate” the microbiome are only cautiously explored in specific human conditions.
Diet connections:
Studies suggest that high-fiber, plant-rich diets associate with more diverse, favorable microbiome profiles. Frequent ultra-processed foods and low fiber intake link with less favorable patterns.
On probiotics and prebiotics: Certain strains and fibers show evidence for specific conditions (antibiotic-associated diarrhea, for instance). Broad “longevity” claims aren’t established.
Supporting microbiome resilience:
Gradually increase dietary fiber from varied sources
Include fermented foods if tolerated
Stay physically active
Limit unnecessary antibiotics (with medical guidance)
Maintain a healthy diet emphasizing whole foods
How the 12 Hallmarks Interact: A Systems View of Aging
The 12 hallmarks of aging don’t operate independently. They form an interconnected network with reciprocal influences and feedback loops.
Example cascade:
Poor sleep and chronic stress → elevated cortisol and sympathetic nervous system activity → increased inflammation and metabolic dysregulation → greater oxidative stress and mitochondrial strain → accelerated damage across multiple hallmarks
This is a conceptual model, not a guaranteed sequence, but it illustrates how foundational behaviors compound into systemic effects over time.
Why this matters:
Single “magic bullet” supplements or interventions rarely address the full picture. The systems perspective explains why multifactorial lifestyle patterns—addressing movement, nutrition, sleep, social health, and toxin avoidance simultaneously—likely produce greater positive effects than targeting any single pathway.
Think in terms of balancing stress and recovery at both cellular and whole-body levels. Small, sustainable changes can compound over decades into meaningful differences in health span.

Evidence-Based Lifestyle Foundations for Healthy Aging
High-quality evidence consistently points to several lifestyle foundations that support healthy ageing. These don’t promise to “reverse” specific hallmarks or guarantee life expectancy gains, but they align with better outcomes across longevity and public health research.
Movement
Movement: Current guidelines recommend 150-300 minutes weekly of moderate aerobic activity plus two strength training sessions. This level of regular physical activity supports mitochondrial function, vascular health, blood vessels elasticity, insulin sensitivity, and muscle mass preservation—addressing multiple hallmarks simultaneously. Even starting in old age shows benefits for reducing death rate from major causes. Being more active may lower your risk of heart attack, stroke, falling, and diabetes. Regular physical activity can also improve your mood and help you sleep better.
Nutrition
Nutrition: Emphasize minimally processed, plant-forward eating patterns with adequate protein (especially important for older adults), limited added sugars, and reduced ultra-processed food intake. Such approaches connect conceptually to nutrient sensing regulation, inflammation reduction, and microbiome health. Think whole grains, vegetables, fruits, legumes, nuts, and quality protein sources. The Mediterranean diet is credited with and associated with lowering the risk of heart disease and early death.
Sleep
Sleep: Consistent 7-9 hours for most adults ties to better metabolic profiles, cognitive function, and inflammatory markers. Sleep deprivation impairs the glymphatic system, disrupts hormone regulation, and accelerates multiple hallmarks. Prioritizing enough sleep is one of the most accessible interventions available.
Mental and Social Health
Mental and social health: Strong evidence shows that social connection, sense of purpose, and stress management practices—mindfulness, hobbies, nature exposure—correlate with lower mortality, better cardiovascular outcomes, and possibly slower functional decline. Maintaining relationships with loved ones and community engagement matters for well being.
Medical Prevention
Medical prevention: Keep up with age-appropriate screenings and vaccinations. Manage blood pressure, cholesterol, and glucose with clinician guidance. Avoid tobacco entirely. These steps reduce the burden of age-related diseases that interact with underlying hallmarks.
Quick reference checklist:
[ ] 150+ minutes moderate activity weekly
[ ] 2 strength training sessions weekly
[ ] 7-9 hours quality sleep nightly
[ ] Whole-food, plant-forward eating pattern
[ ] Regular social connection and purpose
[ ] Up-to-date screenings and vaccinations
[ ] Tobacco-free lifestyle
Note: Age-related hearing loss can affect the ability to hear high pitched sounds, which is a common sensory change with aging.
Emerging Longevity Interventions: Hype vs Cautious Optimism
Several intervention categories are under intense investigation:
| Category | Current Status | Key Considerations |
|---|---|---|
| Senolytics | Early human trials | Promising animal data; side effects under study |
| Metformin (geroprotective) | Large trials ongoing | Already approved for diabetes; aging indication experimental |
| Rapamycin analogs | Research stage | Immune suppression concerns |
| NAD+ boosters | Pilot studies | Long-term human effects unknown |
| Epigenetic reprogramming | Animal studies | Partial reprogramming showing promise in mice |
| Important context: Most promising results come from yeast, worms, flies, and mice—or early human pilot studies. Long-term benefits and risks in diverse human populations remain unknown. | ||
| No regulatory pathway currently exists for approving a general “anti-aging” therapy. Compounds get approved for specific diseases (rapamycin for transplant rejection, metformin for diabetes) rather than for aging itself. |
Be skeptical of products or clinics promising dramatic rejuvenation, biological age reversal, or disease cures without strong peer-reviewed evidence and regulatory oversight.
Questions worth pondering:
How much risk is acceptable for hypothetical life span gains?
How should society prioritize access if interventions prove effective?
What non-pharmaceutical approaches deserve continued emphasis regardless?
Frequently Asked Questions About the Hallmarks of Aging
Can I measure my biological age right now?
Several tests exist—epigenetic clocks, telomere length measurements, biomarker panels. However, these have limitations. Different clocks can give different estimates, and clinical decisions rarely rely on them alone. They’re useful research tools but not yet precise enough to guide individual interventions. Discuss any testing with healthcare providers.
What is the single most effective thing I can do to age well?
There’s no single factor. However, evidence suggests consistently that avoiding smoking, maintaining regular physical activity, and staying at a healthy weight are among the most powerful predictors of good health and longevity in large cohort studies. These address multiple hallmarks simultaneously.
Is aging a disease that can be cured?
This remains debated. Some researchers propose classifying aging as a disease to enable regulatory pathways for treatments. Others view it as a natural, complex process best addressed through multifactorial approaches. Currently, medicine treats diseases of aging—heart disease, cancer, dementia—rather than aging itself.
How soon will we have proven anti-aging drugs?
Timelines remain uncertain. Large trials like the TAME study (Targeting Aging with Metformin) are ongoing, but even positive results would likely complement rather than replace lifestyle and preventive care. Expect gradual progress rather than sudden breakthroughs.
Is it too late to start healthy habits in my 50s, 60s, or later?
Studies suggest that adopting healthy behaviors in midlife and beyond still reduces risk of chronic diseases and disability. Young people certainly benefit from starting early, but the “best time to start” is always now. Earlier adoption yields larger cumulative benefit, but it’s never too late to see improvements.
Should I follow extreme diets or biohacking protocols?
Approach with caution. Extreme interventions carry nutritional and psychological risks, and many lack rigorous human evidence. Evidence-based, sustainable approaches under medical supervision are preferable. Remember that short lifespan interventions in animal models don’t automatically translate to safe, effective human therapies.
Conclusion: Aging as a Scientific and Personal Journey
The 12 hallmarks of aging provide a powerful framework for understanding why bodies change over time. They connect the molecular basis of cellular damage to the visible, functional changes we experience as years pass—from memory loss to reduced energy to increased disease risk.
While cutting-edge interventions are under development, the most reliable paths to healthy ageing currently involve accessible behaviors and preventive care decisions that support multiple hallmarks simultaneously. This isn’t about chasing life extension at any cost—it’s about maintaining independence, cognitive function, and meaningful relationships for as many years as possible.
Aging presents both challenges and opportunities for curiosity. Research from the 2020s continues refining our understanding; the hallmarks framework itself will likely evolve as new discoveries emerge. The National Institute on Aging and research institutions worldwide continue to study aging mechanisms, bringing us closer to practical applications.
Consider discussing what you’ve learned here with your healthcare professionals. Start with one sustainable change aligned with evidence. Recognize that small steps—better sleep, regular movement, nourishing food, genuine connection—can compound into significant long-term benefits.
The science of longevity invites you to participate not just as a passive observer, but as someone who can apply emerging knowledge to support your own health span. That journey begins with understanding—and now you have a foundation in the 12 hallmarks of aging to build upon.

Blood Vessels and Cardiovascular Health: Aging’s Impact on the Circulatory System
The normal ageing process brings about profound changes in the circulatory system, shaping the health of blood vessels and the heart over time. As we age, our bodies accumulate damage to genetic material and cells—a process driven by factors like genomic instability, mitochondrial dysfunction, and cellular senescence. These underlying mechanisms, central to the hallmarks of aging, directly impact cardiovascular health and contribute to the development of age related diseases.
Blood Vessel Changes with Age
One of the most noticeable changes is the stiffening and narrowing of blood vessels. Over the years, blood vessels lose their youthful elasticity, making it harder for them to expand and contract as needed. This stiffening is partly due to the buildup of reactive oxygen species (ROS), which cause oxidative damage to cells and tissues. As a result, older adults are more likely to experience high blood pressure and an increased risk of heart disease.
Heart Structure and Function
The heart itself is not immune to the aging process. Structural and functional changes—such as thickening of the heart walls and reduced pumping efficiency—can develop gradually, raising the risk of heart failure and other age related conditions. Cellular senescence plays a key role here: as cells divide and age, some become senescent, stop dividing, and begin to form aggregates. These senescent cells can disrupt normal tissue function and alter intercellular communication, further contributing to cardiovascular decline.
Genetic and Telomere Effects
Genetic instability and telomere shortening also influence cardiovascular health. Each time cells divide, their telomeres—the protective caps on chromosomes—shorten. When telomeres become too short, cell function declines and the risk of cell death increases. While most cells eventually stop dividing, some cancer cells can express telomerase, allowing them to maintain telomere length and continue dividing indefinitely. Research into telomere lengthening and DNA repair is ongoing, with the hope of finding ways to support healthy ageing and extend lifespan.
Mitochondrial Function and Cardiovascular Health
Mitochondrial function is another crucial factor. As we age, mitochondria—the energy factories of our cells—become less efficient, leading to reduced energy production and increased oxidative stress. This decline in mitochondrial function can impair heart and blood vessel health, making the circulatory system more vulnerable to damage.
Lifestyle Choices for Cardiovascular Health
Fortunately, evidence suggests that lifestyle choices can have a powerful impact on cardiovascular health, even in old age. A healthy diet rich in whole grains, fruits, and vegetables provides antioxidants and nutrients that help combat oxidative stress. Regular physical activity—such as walking, swimming, or weightlifting—supports blood vessel flexibility, improves mitochondrial function, and helps maintain a healthy weight. Getting enough sleep and managing stress are also important for good health and well-being.
Caloric restriction, when done safely and under medical supervision, has shown promise in animal studies for extending lifespan and improving cardiovascular health. While more research is needed to determine the best approach for humans, these findings highlight the importance of balanced nutrition and avoiding extra weight.
The National Institute on Aging and other research organizations continue to study the molecular basis of aging, focusing on areas like cellular senescence, mitochondrial dysfunction, and genetic instability. Their work aims to uncover new strategies to promote healthy ageing and reduce the burden of age related diseases.
In summary, the aging process has a significant impact on blood vessels and cardiovascular health, but individuals can take proactive steps to support their circulatory system. By embracing regular physical activity, following a healthy diet, getting enough sleep, and managing stress, older adults can help maintain good health and well-being well into old age. Ongoing research continues to reveal new ways to extend lifespan and improve human health, offering hope for healthier hearts and longer lives.
Further Reading
Explore more articles related to this topic:
- The Link Between NMN and Telomere Length: Can You Actually Reverse Biological Age?
- NMN Research Update 2026: What Recent Human Trials Tell Us About Reversing Biological Age
- What Are Senolytics? A Beginner’s Guide to How Quercetin and Fisetin Clear “Zombie Cells”
- Best Longevity Supplements in 2026: Science-Backed Picks Only
- NMN and Urolithin A: The New Frontier of Mitochondrial Health in 2026