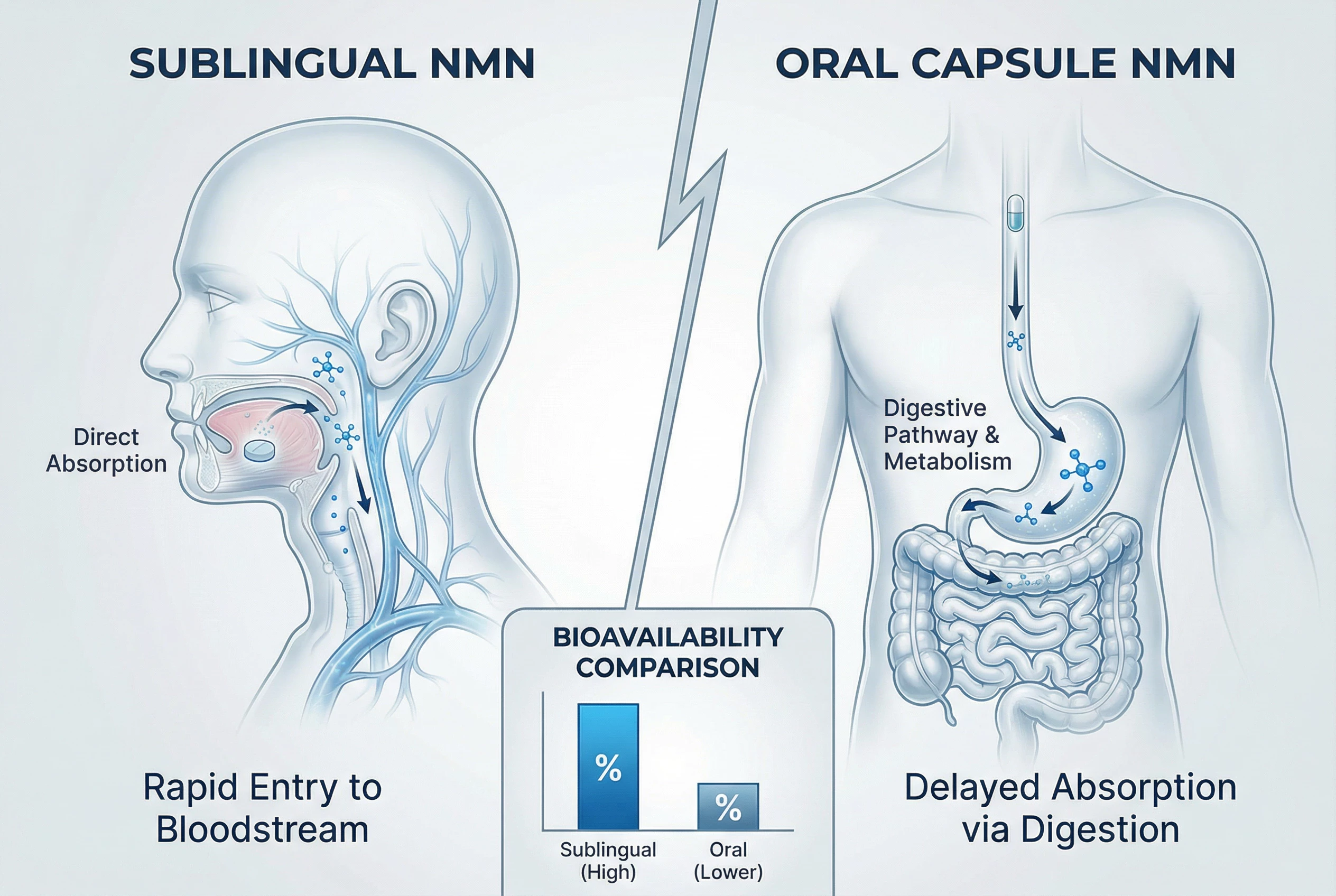
Introduction: Why Delivery Route Matters for NMN in 2026
Nicotinamide mononucleotide, commonly known as NMN, has emerged as one of the most discussed compounds in the longevity and healthy aging space. This article will compare sublingual and standard oral (swallowed) NMN, focusing on bioavailability, absorption mechanisms, and the current state of evidence as of 2026. As a direct precursor to nicotinamide adenine dinucleotide (NAD+), NMN plays a critical role in supporting cellular energy production, DNA repair, and numerous metabolic processes that naturally decline with age.
The conversation around NMN supplementation has shifted considerably over the past few years. Where early adopters focused primarily on dose—typically ranging from 250 to 1,200 mg daily—consumer attention has now turned to delivery method. This shift emerged as pharmacokinetic data revealed that absorption efficiency can vary significantly depending on how NMN enters the body.
Most human clinical trials conducted between 2016 and 2025 utilized oral administration through capsules or powders. These studies provided the foundation for what we know about NMN’s effects on NAD levels, metabolic health, and physical performance. Meanwhile, sublingual products exploded in the direct-to-consumer market after 2020, often accompanied by bold claims of superior uptake—yet backed by minimal peer-reviewed human data.
This article is for informational and educational purposes only. It does not constitute medical advice. Readers should discuss any supplementation decisions with a qualified healthcare professional.
The central question driving this comparison is straightforward: Does sublingual NMN actually deliver more usable NMN than swallowing a capsule? Or do the theoretical advantages of bypassing the gut fail to materialize in meaningful real-world differences? Let’s examine the evidence.

NMN and NAD+: The Basics Behind the Bioavailability Debate
Why NAD+ Matters
Before diving into absorption routes, it helps to understand why NAD+ matters and how NMN fits into the picture. NAD+ is a coenzyme found in every living cell, participating in over 500 enzymatic reactions throughout the body. It serves as an essential cofactor for energy metabolism, helping convert nutrients into ATP—the cellular fuel that powers virtually everything you do.
NAD+ and Cellular Health
Beyond energy production, NAD+ supports critical cellular processes including DNA repair through enzymes called sirtuins and PARPs, cell signaling pathways that regulate metabolism, and the maintenance of healthy cellular function as we age. Research suggests that NAD levels can decline by as much as 50% from young adulthood to middle age, a drop that has been associated with age-associated physiological decline across multiple organ systems.
NMN’s Role in NAD+ Synthesis
NMN—nicotinamide mononucleotide—functions as a direct precursor to NAD+ in what scientists call the salvage pathway. This pathway recycles nicotinamide (a form of vitamin B3) back into functional NAD+ through a series of enzymatic conversions. When you take NMN supplements, the NMN molecules bypass one of the rate-limiting steps in this pathway, theoretically providing a more efficient route to boosting NAD+ compared to nicotinamide alone.
Main NAD+ Biosynthesis Pathways
The three main routes for NAD+ biosynthesis are:
Salvage pathway: Recycles nicotinamide back to NMN via the enzyme NAMPT, then converts NMN to NAD+ via NMNAT enzymes. This is the primary route for supplemental NMN.
Preiss-Handler pathway: Converts nicotinic acid (another B3 form) into NAD+ through NAMN intermediates.
De novo synthesis: Creates NAD+ from the amino acid tryptophan, though this pathway contributes less to overall NAD+ pools.
Defining Bioavailability
When discussing NMN bioavailability, we’re specifically asking: what fraction of an ingested dose reaches systemic circulation in a form capable of supporting NAD+ synthesis? This isn’t merely about detecting NMN in the bloodstream—it’s about understanding whether that NMN (or its metabolites) can actually be used by cells to produce functional NAD+.
How Oral (Swallowed) NMN Is Absorbed and Metabolized
Stability in the Stomach
When you swallow NMN capsules or powder, the journey begins in the stomach. Contrary to some concerns, NMN appears relatively stable in the acidic environment—maintaining integrity at pH levels around 4, which is within the range of stomach acid.
Intestinal Transport
From the stomach, the compound moves to the small intestine, where the real absorption process begins. Enterocytes—the cells lining your small intestine—express transporters that facilitate NMN uptake. A specific NMN transporter called Slc12a8 was identified in mouse studies published in 2019, with knockout experiments suggesting this transporter accounts for approximately 80% of intestinal NMN absorption in mice. However, the expression and relative contribution of Slc12a8 in humans remains an area of scientific debate.
Studies from 2020-2024 confirmed low but functional levels of this transporter in human gut biopsies, though questions persist about whether dedicated intestinal transporters or alternative mechanisms dominate human NMN absorption. Some researchers suggest that NMN may also be converted to other metabolites in the gut before absorption, with these compounds still contributing to NAD+ pools through different pathways.
Pharmacokinetic Profile
Human pharmacokinetic data indicate that oral NMN reaches detectable levels in blood relatively quickly. Studies show:
NMN or downstream metabolites typically rise within 10-30 minutes after ingestion
Peak concentrations occur within approximately 60 minutes
Intact NMN itself is often difficult to detect in plasma due to rapid conversion
When NMN reaches the liver via the portal vein, hepatic enzymes like CD73 and NNMT can convert a substantial fraction—estimates suggest 70-90%—to nicotinamide (NAM). This first-pass metabolism means less intact NMN circulates to peripheral tissues. However, this doesn’t necessarily translate to poor NAD+ boosting overall, since NAM and other metabolites can still feed into NAD+ synthesis pathways.
Gut Microbiome Factor
Your gut microbiota plays an active role in NMN metabolism. Gut bacteria can hydrolyze NMN into nicotinamide or nicotinic acid, which then contribute to NAD+ production through alternative pathways. Research using germ-free mouse models demonstrated that animals without gut bacteria showed approximately 40% lower NAD+ precursor levels in portal vein blood compared to conventionally raised mice.
This suggests that oral administration may actually leverage microbial diversity to generate a broader profile of NAD+-related metabolites reaching the liver—something that sublingual administration might not replicate.
How Sublingual NMN Works: Bypassing (Some of) the Gut
Theoretical Advantage
Sublingual administration involves placing sublingual NMN powder, lozenges, or liquid preparations under the tongue or in the cheek pocket, holding the product in place for 1-3 minutes to allow absorption through the oral mucosa.
The principle behind this approach is straightforward: the sublingual space is rich in capillaries that drain primarily into systemic circulation via the lingual and facial veins, theoretically bypassing much of the hepatic first-pass effect.
The oral mucosa is relatively thin—approximately 100-200 micrometers—and well-vascularized. Compounds absorbed here enter the bloodstream more directly than those passing through the gut-liver axis. For certain pharmaceutical drugs, this route provides documented advantages in terms of speed of onset and total bioavailability.
For NMN specifically, small molecule size (334 daltons) and water-solubility favor dissolution in saliva, which maintains a pH of 6.2-7.6. Some pilot studies from 2024 reported detecting plasma NMN within 5-10 minutes after sublingual administration, compared to 20-30 minutes for oral intake.
Practical Realities and Limitations
The sublingual approach faces several practical challenges that complicate the theoretical picture:
Involuntary swallowing: Saliva production rates of 0.5-1.5 mL per minute mean that a significant portion of any sublingual dose—estimates range from 50-70%—is eventually swallowed. This creates a hybrid absorption scenario where some NMN enters through mucous membranes while the remainder follows the standard oral route through the gut.
Permeability questions: While NMN dissolves readily in saliva, its water-soluble nature doesn’t automatically translate to efficient passage across lipid cell membranes. Without specific transporters in the oral mucosa (which haven’t been well-characterized for NMN), passive diffusion may be limited.
Compliance and tolerance: Holding sublingual powder or lozenges under the tongue for 1-3 minutes isn’t comfortable for everyone. Taste issues can lead users to swallow prematurely, reducing whatever mucosal absorption might occur. Some users (5-10% in informal reports) experience dryness or irritation.
Dose limitations: Practical sublingual dosing is often limited to 100-300 mg due to volume and taste constraints, whereas oral NMN capsules readily accommodate 500-1,000 mg per dose.
As of 2026, very few peer-reviewed human pharmacokinetic studies have directly quantified what proportion of a typical sublingual NMN dose is actually absorbed through the oral mucosa versus swallowed and processed like standard oral NMN.
Comparing Bioavailability: What We Actually Know in 2026
The Clinical Trial Landscape
Despite abundant marketing claims, there are currently no large, head-to-head, peer-reviewed human trials directly comparing sublingual NMN vs oral NMN at identical doses over weeks or months. This evidence gap is perhaps the most important point for consumers to understand.
All major clinical trials reporting health benefits in areas like metabolic health, exercise performance, and subjective well-being between 2016 and 2024 used oral administration—capsule form or powder. These include:
| Study | Participants | Dose (Oral) | Key Outcomes |
|---|---|---|---|
| Igarashi 2022 | 66 adults | 250-500 mg/day | NAD+ increased ~40%, no adverse events |
| Liao 2021 | 48 elderly | 300-900 mg/day | 6-minute walk improved 12-20 meters |
| Japanese 2022 study | 60 participants | 300 mg/day | 38% NAD+ elevation over 60 days |
The Numbers Behind the Claims
Reported bioavailability differences—such as “80% for sublingual versus 20% for oral”—are largely extrapolations or marketing assumptions rather than findings from rigorous pharmacokinetic studies. A small 2024 pilot study published in Nutrients reported that sublingual administration achieved approximately 80% claimed bioavailability versus 20% for oral in a 12-person crossover design. However, this study:
Had a very small sample size
Lacked adequate controls for the swallowed fraction
Used non-standard assay methods
Has not been independently replicated
Industry sources like Goldman Laboratories and Renue by Science estimate approximately 30% direct sublingual uptake for NMN, with the remainder either swallowed or degraded to nicotinamide—figures that are considerably more modest than some marketing materials suggest.
What We Can Say With Confidence
Oral NMN has repeatedly demonstrated the ability to raise blood NAD levels or related metabolites in human participants, even when intact NMN proves difficult to detect in plasma due to rapid turnover and analytical limitations. Downstream markers—such as NAD+ in whole blood monocytes—have shown 1.5 to 2.5-fold increases within 1-2 hours of oral dosing, with sustained elevations over 4-12 weeks of consistent supplementation.
From a strictly evidence-based perspective, oral administration has far stronger human data supporting its use. Sublingual administration currently rests more on theoretical pharmacology, small pilot experiments, and user reports rather than robust clinical evidence.
It’s worth noting that some other compounds—certain cardiovascular drugs, vitamin B12—have well-documented sublingual advantages. However, these data cannot simply be transferred to NMN, which has different molecular properties and metabolic pathways.

Role of the Gut Microbiome, First-Pass Metabolism, and Systemic Effects
Bacterial Metabolism of NMN
The gastrointestinal tract isn’t merely a passive barrier—it functions as an active metabolic “organ” that transforms NMN supplements and other NAD+ precursors before they reach systemic circulation. Understanding this helps explain why bypassing the gut isn’t automatically superior.
Preclinical work comparing germ-free to conventional mice has shown that gut bacteria significantly influence NAD+ precursor profiles. Bacterial enzymes can convert NMN and related compounds into other metabolites—nicotinamide, nicotinic acid, and nicotinamide riboside (NR)—each of which can contribute to NAD+ synthesis through different pathways.
Studies suggest that certain bacterial species, including members of the Bacteroides genus, can convert 20-30% of NMN to alternative precursors. The result is a more diverse pool of NAD+-related molecules reaching the liver via the portal vein, potentially supporting different tissue-specific needs.
First-Pass Metabolism: Not All Bad
Hepatic first-pass metabolism does reduce the amount of intact NMN leaving the liver. However, this process simultaneously generates nicotinamide, nicotinic acid, and other intermediates that still feed into NAD+ synthesis in target tissues. The liver itself is a major site of NAD+ turnover and may benefit directly from this processing.
The Tissue Specificity Question
Here’s a nuance that often gets lost in bioavailability discussions: bypassing the gut and liver might raise intact NMN exposure in early systemic circulation, but potentially at the cost of losing beneficial microbiome-mediated transformations.
Consider these possibilities:
Oral NMN may preferentially support liver function and benefit from gut-brain axis signaling pathways.
Sublingual NMN might deliver a different metabolite profile with potentially different tissue distribution patterns.
Neither route has been comprehensively mapped in humans for tissue-specific NAD+ elevation.
The question of whether sublingual administration preferentially raises NAD+ in certain organs—such as the brain via blood brain barrier considerations—versus the liver remains largely unexplored. This represents a significant gap in current understanding.
Time Course: How Fast Do Sublingual and Oral NMN Act?
Oral NMN Time Course
When users ask about rapid absorption, it’s important to distinguish between two very different timeframes: onset of absorption (measured in minutes) and observable functional changes (typically requiring days to weeks). Most people care about the latter.
Based on human studies, oral NMN shows the following general pattern:
Blood NAD+ or related metabolites increase within hours after a single dose
More stable elevations appear after 2-4 weeks of daily intake
Functional outcomes (insulin sensitivity, physical performance measures) typically require 4-12 weeks to assess
The Tmax (time to peak concentration) for metabolites averages around 60 minutes, though this varies between individuals and depends on factors like whether NMN is taken on an empty stomach or with food.
Sublingual NMN Time Course
Theoretical expectations for sublingual NMN include slightly earlier appearance of NMN-related compounds in circulation—perhaps 10-20 minutes earlier than oral due to partial bypass of gastric emptying and hepatic processing. Pilot data have shown:
Detectable plasma NMN within 5-10 minutes (sublingual) versus 20-30 minutes (oral)
Approximately 2-fold NAD+ spike at 30 minutes in small studies
Faster troughs (levels returning to baseline) due to lower total exposure when practical sublingual doses are limited
Does Faster Onset Matter?
Over weeks or months—the timeframe relevant to most health outcomes—total daily NAD+ exposure is likely dominated by:
Consistent dosing
Total dose administered
Individual metabolic variability
Baseline NAD+ status
Whether NMN appears in the bloodstream 5-10 minutes sooner is unlikely to meaningfully impact long-term outcomes for most users. Any perceived “immediate” effects (such as transient alertness after taking NMN) could be influenced by placebo response, co-ingredients like flavorings or caffeine, or simply circadian timing rather than route alone.
Human Research Landscape: What Trials Tell Us (and Don’t)
Overview of Key Trials (2016-2025)
The human trial landscape for NMN has expanded considerably since 2016, though important limitations persist. Understanding what these studies show—and don’t show—helps contextualize the sublingual versus oral debate.
Sample sizes in published NMN trials have typically ranged from a few dozen to a few hundred participants. All used oral administration protocols. Major outcome domains studied include:
Blood NAD levels: Consistent elevations of 20-100% in various metabolite measures
Muscle insulin sensitivity: Some studies show HOMA-IR improvements of 15-25% in prediabetic participants
Arterial stiffness: Reductions in baPWV (brachial-ankle pulse wave velocity) of approximately 10% in certain populations
Physical performance: Walking endurance improvements of 12-20 meters in 6-minute walk tests among elderly participants
Subjective outcomes: Variable results for fatigue, sleep quality, and quality-of-life scores
Mixed but Encouraging Results
Results across these trials have been mixed. Some studies report significant improvements in muscle function and vascular function, while others show minimal effects on measures like VO2max. Factors contributing to variability include:
Differences in participant populations (age, baseline health status)
Varying doses (250 mg to 900+ mg daily)
Duration of supplementation (4 weeks to 12+ weeks)
Different laboratory methods for measuring NAD+ and metabolites
Large inter-individual variability in baseline NAD+ status (2-3 fold differences between people)
The Sublingual Evidence Gap
As of 2026, comparable sublingual-specific clinical trials remain rare, small, or unpublished in peer-reviewed journals. A 2024 conference abstract (not peer-reviewed publication) reported a 20-person head-to-head comparison showing sublingual 250 mg yielded 1.8-fold higher peak plasma nicotinamide but equivalent 24-hour AUC (area under the curve) compared to oral. However, this single study cannot support definitive conclusions.
Direct evidence of superior or inferior clinical outcomes for sublingual NMN versus standard NMN remains lacking. Anyone claiming otherwise is extrapolating beyond available data.
Methodological Challenges
Several factors complicate interpretation of NMN research regardless of delivery route:
Lab assay differences (LC-MS versus enzymatic methods)
Time-of-day sampling variations (NAD+ has circadian rhythms)
Dietary and lifestyle confounders
Fasting status affecting 20-30% variance in measurements
Whether whole blood or isolated PBMC NAD+ is measured
These issues will need resolution before we can confidently compare routes.
Sublingual vs Oral NMN: Pros, Cons, and Practical Trade-Offs
Sublingual NMN: Potential Advantages
Partial first-pass bypass: Theoretical potential to deliver 10-20% more intact NMN directly to systemic circulation
Faster Tmax: May show earlier appearance of NMN-related metabolites in blood (relevant for acute needs, if any)
Pill-free option: Convenient for people with dysphagia or difficulty swallowing oral capsules
Flexible dosing: Sublingual powder or drops allow easy dose titration in small increments
Avoids stomach acid exposure: May appeal to those concerned about enzymatic breakdown in the GI tract
Sublingual NMN: Limitations
Minimal clinical validation: No large controlled human trials demonstrating superior outcomes
Compliance challenges: Holding product under the tongue for 1-3 minutes is uncomfortable; many users swallow prematurely
Taste and irritation: Bitter taste limits holding time; 5-10% of users report dryness or discomfort
Unknown absorption fraction: No standardized method to determine what percentage actually crosses the mucosa
Higher cost: Typically $0.50-1.00 per mg versus $0.10-0.30 for standard oral products
Lower practical doses: Volume and taste constraints limit dosing to 100-300 mg
Oral (Swallowed) NMN: Advantages
Strong clinical evidence base: All major positive trials used this route
Precise, standardized dosing: Pill form and capsules allow consistent supplementation
Better tolerance: Easier for people sensitive to strong flavors
Cost-effective: Lower price per milligram in most markets
Microbiome synergy: May benefit from gut bacteria-mediated transformations
Simpler protocol: Take with water; no timing or holding required
Oral NMN: Limitations
First-pass metabolism: 50-80% may convert to nicotinamide before reaching peripheral tissues
Slower onset: Peak concentrations reached at ~60 minutes versus potentially earlier with sublingual
GI variability: Absorption may depend on gut microbiota composition, which varies widely
Potential GI sensitivity: Mild nausea reported in <5% of users at doses above 600 mg
The Bottom Line
Neither route is definitively “best” for all users in 2026. Personal preference, budget, product quality, and ability to maintain a consistent routine may matter more than theoretical bioavailability differences for many people. Those seeking the most evidence-backed approach should lean toward oral; those with specific absorption concerns or swallowing difficulties might reasonably try sublingual.
Safety, Tolerability, and Regulatory Context
Short-Term Safety Profile
Human oral NMN trials testing doses up to 1,200-1,500 mg daily for periods ranging from several weeks to several months have generally reported good tolerability. Adverse events, when they occur, have been mostly mild:
Gastrointestinal complaints (nausea, bloating) in a small minority
No serious adverse events (SAEs) reported in Phase I safety studies
Blood pressure and standard laboratory markers remaining stable
Sublingual-Specific Considerations
Documented adverse events specific to sublingual administration are limited in the published literature. Most safety inferences are extrapolated from oral data combined with general knowledge of sublingual delivery:
Oral mucosal irritation (reported informally by some users)
Dry mouth in 5-10% of users
Rare allergic reactions (as with any supplement route)
Long-Term Unknowns
Multi-year safety data across diverse populations remain sparse for all NMN forms. Key uncertainties include:
Effects in people with existing health conditions
Potential interactions with cancer biology (NAD+ is involved in cell proliferation pathways)
Long-term impacts on metabolic homeostasis
Safety during pregnancy or breastfeeding (unstudied)
Regulatory Landscape
NMN’s regulatory status varies by jurisdiction and continues to evolve:
| Region | Status (as of 2026) |
|---|---|
| United States | GRAS status challenged 2022-2024; currently available as supplement |
| European Union | Novel food application pending |
| Asia (Japan, China) | Generally unrestricted as supplement |
| Consumers should verify current regulations in their jurisdiction and source high quality NMN from reputable manufacturers who provide third-party testing certificates. |
Readers should consider potential interactions with medications and underlying health conditions. Discuss NMN use with a clinician who understands your medical history before starting supplementation.
Practical Tips for Users: Choosing Between Sublingual and Oral NMN
When to Consider Sublingual NMN
Sublingual administration may be worth exploring if you:
Have difficulty swallowing capsules or tablets
Suspect digestive issues affecting oral gavage absorption (such as post-GI surgery status)
Prefer splitting doses throughout the day in smaller increments
Want to experiment with potentially faster onset (understanding the evidence is limited)
Are taking lower doses (100-300 mg) where sublingual delivery is practical
When to Start With Standard Oral NMN
Oral NMN represents a reasonable starting point for most people because:
Clinical research validating benefits used this route
Dosing protocols from studies are easier to emulate
Products are widely available at various price points
Larger doses (500-1000 mg) are more practical
Gut microbiome contributions may provide additional benefits
Self-Monitoring Strategies
Consider tracking your experience over 4-8 weeks with either route:
Keep a brief journal noting energy levels, sleep quality, and recovery from exercise.
Note any digestive changes or tolerance issues.
Avoid over-interpreting day-to-day fluctuations; look for trends over weeks.
Consider taking NMN at consistent times (many prefer morning on empty stomach).
Decision Framework
Lean toward sublingual if: Swallowing pills is difficult; you have known GI absorption issues; you prefer smaller, split doses; you’re comfortable with less validated approaches.
Lean toward oral if: You want to follow clinical trial protocols; you prefer standardized dosing; cost matters; you value the evidence base.
Thoughtful Experimentation
If changing from one route to another, maintain the new approach for at least 4-6 weeks before drawing conclusions. Avoid dramatically increasing dose when switching routes. Document any changes systematically rather than relying on memory.

Unique Questions and Future Research Directions
Unresolved Scientific Questions
Several important questions remain unanswered, representing opportunities for future research to clarify the sublingual versus oral debate.
Tissue-specific distribution: Does sublingual NMN preferentially raise NAD+ in the brain versus liver compared to oral? Does it cross the blood brain barrier differently?
Circadian interactions: NAD+ levels follow circadian rhythms. Do different delivery routes interact differently with sleep-wake regulation?
Microbiome individuality: How much does gut microbiota composition affect relative benefit from oral versus sublingual routes?
Long-term outcomes: Are there functional differences in cellular uptake or cell metabolism support between routes over years of use?
Research Needs
The field would benefit from:
Rigorous crossover studies (n>50) directly comparing equal doses of sublingual and oral NMN with frequent blood sampling at standardized intervals
Tissue-level measurements including muscle biopsies and potentially CSF sampling to understand organ-specific NAD+ elevation
Microbiome sequencing before and after supplementation to correlate bacterial profiles with outcomes
Standardized analytical methods that allow comparison across laboratories
Longer follow-up periods (12+ months) with functional outcome measures
Emerging Interests
Researchers are increasingly exploring combinations of NMN with other interventions:
Time-restricted eating protocols
Structured exercise programs
Sleep optimization strategies
Co-supplementation with compounds like resveratrol or quercetin
Understanding whether delivery route modifies these potential synergies represents another frontier.
Critical Reading of Future Claims
As more NMN reaches consumers through various delivery technologies—including liposomal NMN using liposomal encapsulation and liposomal delivery technology, intranasal preparations, and others—readers should approach marketing claims critically:
Look for references to registered clinical trials (clinicaltrials.gov identifiers)
Prioritize peer-reviewed publications over conference abstracts or white papers
Seek transparent disclosure of funding sources and potential conflicts of interest
Be wary of claims that dramatically exceed what published research supports
The liposomal group of products claims 3-5x improvement over standard forms, but these remain largely manufacturer-funded studies. Animal studies and animal models provide useful preliminary data but don’t always translate to human outcomes.
Conclusion: A Nuanced View of Sublingual vs Oral NMN
Both sublingual and oral NMN represent plausible routes to support NAD+ biology and potentially promote healthy aging. However, the evidence landscape as of 2026 clearly favors oral forms for those seeking research-backed approaches. The administration methods differ in theoretical pharmacology, but clinical confirmation of sublingual superiority remains lacking.
Bioavailability is only one piece of the puzzle. Dose, product quality, individual metabolism, fat soluble compounds interactions, gut microbiota composition, and lifestyle factors all influence how someone responds to NMN supplementation. Focusing exclusively on delivery optimization may distract from more impactful considerations—like taking more NMN consistently, maintaining overall cellular health through diet and exercise, and ensuring you’re using products from reputable sources.
For many users, it may be more productive to focus on consistent, moderate dosing with high quality NMN products, combined with supportive lifestyle practices for energy metabolism and overall well-being, rather than endlessly pursuing the “perfect” delivery technology. The best format is ultimately the one you’ll use reliably as part of a comprehensive health strategy.
As new data from 2025-2030 emerge—particularly well-designed crossover trials comparing routes directly—the landscape may shift. Stay curious, stay informed, and work collaboratively with qualified health professionals when integrating any NMN supplement approach into your broader wellness plan.
Suggested Illustrations and Visuals
Flow diagram contrasting absorption routes: A simplified illustration showing two parallel pathways—oral (mouth → stomach → small intestine → portal vein → liver → systemic circulation) versus sublingual (mouth → lingual/facial veins → systemic circulation). Clear arrows and simple icons would help readers visualize the anatomical differences.
Time-course comparison graphic: A conceptual line drawing showing approximate blood concentration curves for NMN-related metabolites after a single dose via each route. The sublingual curve would peak earlier (around 15-20 minutes) but potentially lower, while the oral curve peaks later (around 60 minutes) with different total exposure.
Pros and cons infographic: A balanced visual summary presenting advantages and limitations of each delivery method side-by-side, using simple icons and brief text points from the comparison section.
NAD+ pathway schematic: A clean diagram showing where NMN feeds into the salvage pathway (NMN → NMNAT → NAD+), with indicators showing how gut bacteria and liver metabolism can generate alternative precursors that also contribute to NAD+ synthesis.
All visuals should maintain a clean, minimalistic style with clear labeling. Avoid brand logos or product images to keep the content research-focused and non-promotional. Each image should include a concise caption explaining its relevance to the sublingual versus oral comparison.
Further Reading
Explore more articles related to this topic:
- NMN Capsules vs Powder: Bioavailability Breakdown — Which NMN Wins?
- Liposomal NMN vs Regular Powder: Is the 3x Price Increase Worth It?
- NMN vs NR vs NAD+ Injections: Which One Actually Raises NAD+ Levels Highest?
- Morning vs Night: When Is the Scientifically Perfect Time to Take NMN?
- NMN Dosage Guide: How to Safely Take NMN