If you’ve been researching ways to protect your vision as you age, you’ve likely come across nicotinamide mononucleotide (NMN) for eye health as a supplement gaining attention in longevity circles. The core topic here is whether NMN can actually help prevent or slow conditions like macular degeneration and vision loss.
This article is for anyone interested in protecting their vision as they age, especially those curious about the potential of NMN supplements for eye health. We’ll cover what NMN is, how it might affect the retina, what current research shows, and what practical steps you can take for eye health. The rising prevalence of age-related eye diseases makes this topic especially important, as more people seek evidence-based strategies to maintain their vision and quality of life. With growing interest in NMN supplements, understanding the science behind their potential role in eye health is crucial.
Worldwide, the global prevalence of age-related eye diseases is rising, and disease burden projection studies highlight the growing future impact of conditions like macular degeneration and vision loss on public health and healthcare systems.
The short answer is that we don’t know yet—at least not with the kind of certainty that would allow doctors to recommend it. But the longer answer reveals a genuinely interesting area of scientific investigation that’s worth understanding.
Answering the Key Question: Can NMN Help Protect Your Eyes?
Let’s address what you came here to learn. Current evidence for NMN and eye health comes mainly from animal and cell studies published between 2018 and 2024. These include experiments using models of retinal detachment, age related macular degeneration, and diabetic retinopathy. Large human clinical trials specifically examining NMN’s effects on vision? Those don’t exist yet.
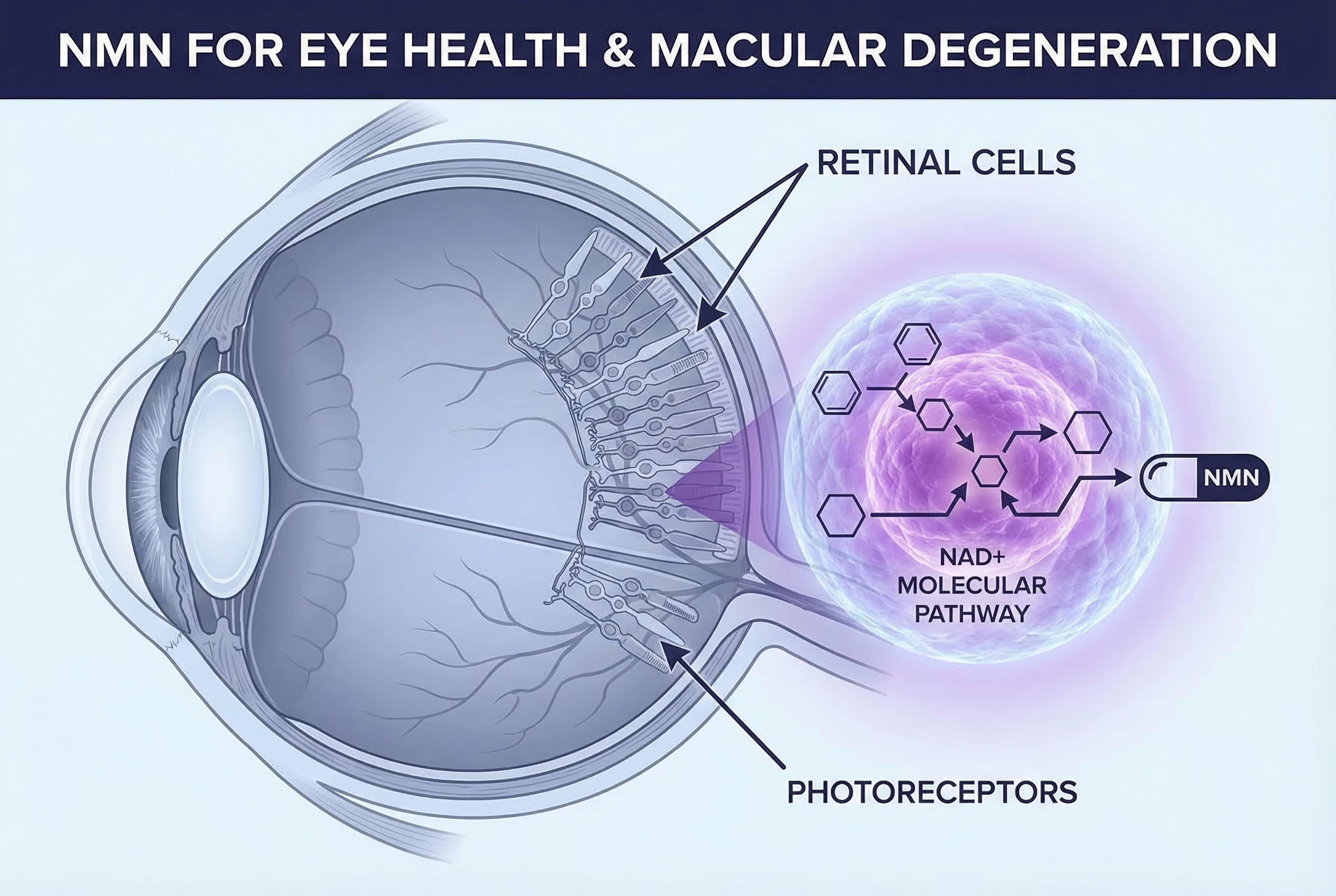
What the research does show is that NMN helps the body produce nicotinamide adenine dinucleotide (NAD+), commonly called NAD+. This molecule is essential for energy production in your retinal cells and for cellular repair mechanisms. When NAD+ levels decline—as they do naturally with aging—retinal cells become more vulnerable to damage.
In addition to macular degeneration, NMN is being investigated for its potential benefits across various ocular diseases and metabolic diseases, due to its role in addressing metabolic dysregulation, inflammation, and mitochondrial dysfunction that are common in these conditions.
In laboratory settings, NMN treatment has demonstrated neuroprotective effects in retinal cells, reducing oxidative stress and inflammation while decreasing photoreceptor cell death. These are promising signals. They suggest that NMN targets the right biological pathways—the same ones implicated in age-related retinal degeneration.
Key Takeaway: Research is promising for neuroprotection of retinal cells in animal models, but it is too early to claim that NMN prevents or treats macular degeneration or vision loss in humans.
However—and this is crucial—demonstrating effects in mice and cell cultures is very different from proving benefit in human eyes. The dosing, delivery methods, and disease models used in experiments don’t translate directly to someone taking an oral supplement.
If you’re experiencing vision symptoms or have been diagnosed with any eye disease, please consult an ophthalmologist before changing your supplements or medications. This article provides educational information, not medical advice.
What This Means for You: NMN may eventually find a place in eye health strategies, but that place hasn’t been established through clinical evidence yet. Consider it an area to watch, not a proven intervention.
What Is NMN and Why Does It Matter for Your Eyes?
Nicotinamide mononucleotide (NMN) is a precursor of nicotinamide adenine dinucleotide (NAD+), which is essential for cellular functions. NMN is synthesized from nicotinamide (NAM) by the rate-limiting enzyme, nicotinamide phosphoribosyltransferase (NAMPT), and is converted into NAD+ by NMN adenylyltransferases (NMNATs). NAD+ is crucial for cellular functions and energy metabolism in the retina. NAD+ levels are known to decline with age, and this decline is linked to the development of pathological retinal conditions such as age-related macular degeneration (AMD) and diabetic retinopathy (DR).
Nicotinamide mononucleotide is a bioactive molecule naturally found in small amounts in foods like broccoli, avocados, and edamame. It’s also produced in your body from vitamin B3-related precursors. Chemically, NMN is a nucleotide—a building block composed of ribose, nicotinamide, and a phosphate group.
What makes NMN significant is its role as a direct precursor to nicotinamide adenine dinucleotide, or NAD+. This coenzyme participates in enormous cellular functions across virtually every tissue in your body. NMN supports cell metabolism (cell metab) by fueling metabolic pathways essential for retinal health, and NAD+ acts as a metabolic sensor activated in response to cellular stress and energy demands. It’s involved in:
Transferring electrons during metabolic reactions that produce cellular energy
Serving as a substrate for enzymes critical to DNA repair
Regulating proteins called sirtuins that influence cellular stress responses
Why NAD+ Decline Matters for Your Retina
NAD+ levels naturally decrease as we age. This decline has been documented in multiple tissues, including the retina in animal studies. The retina is particularly vulnerable to NAD+ deficiency because photoreceptor cells are among the most metabolically demanding cells in the entire body. They require constant ATP production to maintain the ion gradients needed for visual signal processing.
When retinal NAD+ falls, several things can go wrong:
| Consequence of NAD+ Decline | Impact on Retinal Health |
|---|---|
| Reduced mitochondrial function | Less energy for photoreceptors |
| Impaired sirtuin activity | Weakened stress responses |
| Compromised DNA repair | Accumulated cellular damage |
| Increased oxidative stress vulnerability | Accelerated degeneration |
| Research in mouse models has shown that NAD+ deficiency impairs the tricarboxylic acid (TCA) cycle, glycolysis, and SIRT3 activity in retinal tissue. This creates a metabolic insufficiency that may contribute to age related retinal degeneration. |
How NMN Fits Into This Picture
NMN bypasses certain enzymatic steps in NAD+ synthesis, making it a potentially efficient route for boosting NAD+ levels. When you take NMN—whether orally or through other routes—your body can convert it into NAD+ through the enzyme nicotinamide mononucleotide adenylyltransferase.
The key insight for eye health is this: if declining NAD+ contributes to retinal vulnerability, then restoring NAD+ through precursors like NMN might theoretically support retinal function. This is the hypothesis researchers are testing in laboratory settings.
Most mechanistic insights about NMN and the retina come from rodent and cell culture experiments rather than from human trials. This distinction will be important throughout our discussion.
How Aging and Common Eye Diseases Affect the Retina
To understand why NMN might matter for eye health, you need to understand what happens to the retina as we age and how common eye diseases cause damage. As we get older, the retina undergoes changes that can lead to vision decline. This process, known as ocular aging, is driven by molecular mechanisms such as mitochondrial dysfunction, oxidative stress, and cellular senescence, all of which contribute to age-related ocular deterioration.
The Retina and Macula: A Brief Anatomy
The retina is a thin layer of neural tissue—approximately 0.1 to 0.2 millimeters thick—that lines the back of your eye. It contains multiple layers of specialized cells:
Photoreceptors (rods and cones) that convert light into electrical signals
Bipolar cells that process and relay these signals
Ganglion cells whose axons form the optic nerve
Retinal pigment epithelium (RPE) that supports photoreceptor function
Proper retinal development is essential for the formation and maturation of these specialized cells and regions. Disruptions in retinal development can contribute to various retinal disorders.
The macula is a specialized region within the retina, roughly 5 to 6 millimeters in diameter, centered on the fovea. This area contains the highest concentration of cone photoreceptors and is responsible for your central, detailed vision—the vision you use for reading, recognizing faces, and driving.

What Happens to Retinal Tissue with Age
Aging subjects retinal tissue to cumulative stressors that gradually compromise function. The retina experiences particularly high oxidative stress because:
Photoreceptors consume significant oxygen to generate energy
The retina is directly exposed to light, which can generate reactive oxygen species (ROS)
Antioxidant defenses decline with age while ROS production increases
This creates a metabolic imbalance. Additionally, cells can enter a state called cellular senescence—they become dysfunctional but don’t die. Instead, senescent cells persist and secrete pro-inflammatory mediators that can damage surrounding tissue.
Mitochondrial dysfunction compounds these problems. As mitochondria become less efficient, they produce less ATP and more damaging free radicals. This creates a cycle of declining energy production and accumulating damage.
Age-Related Macular Degeneration
Age related macular degeneration is a leading cause of vision loss in people over 60 in developed nations. The disease affects tens of millions of people globally and comes in two forms:
Dry (Atrophic) AMD: Characterized by the gradual accumulation of drusen (lipid-protein deposits) beneath the RPE and progressive atrophy of retinal pigment epithelial cells. This form accounts for roughly 80-90% of AMD cases.
Wet (Neovascular) AMD: Involves abnormal blood vessel growth beneath the retina. These fragile vessels can leak fluid and blood, causing rapid and severe vision loss. Though less common, wet AMD is responsible for most AMD-related blindness.
Both forms ultimately result in photoreceptor death in the macular region, destroying central vision while typically sparing peripheral vision.
Diabetic Retinopathy
Diabetic retinopathy represents a microvascular complication of long-standing diabetes. High blood sugar damages the small blood vessels that supply the retina, leading to:
Microaneurysms and hemorrhages
Macular edema (fluid accumulation)
Eventually, abnormal blood vessel proliferation (proliferative diabetic retinopathy)
The condition also involves neurodegeneration—damage to retinal ganglion cells and other neurons that can precede visible vascular changes. Metabolic dysregulation and complex metabolic dysregulation in diabetes create conditions where NAD+-related pathways may be particularly relevant. Similar mechanisms, such as inflammation and mitochondrial dysfunction, are implicated in various metabolic diseases, highlighting the interconnectedness of metabolic dysregulation in both eye disorders and systemic conditions.
Other Conditions Involving Photoreceptor Degeneration
Several other conditions involve photoreceptor degeneration where metabolic stress and NAD+-related pathways appear relevant:
Retinal detachment: Physical separation of the retina from underlying tissue, causing photoreceptor death. Induced photoreceptor degeneration is often studied in retinal detachment models to better understand mechanisms of injury and test neuroprotective strategies.
Inherited retinal dystrophies: Genetic conditions leading to progressive photoreceptor loss
Light induced retinal degeneration: Damage from excessive light exposure
Ischemic retinopathy: Damage from inadequate blood flow
Treating photoreceptor degeneration remains a major challenge, as there are currently few effective pharmacotherapies for these retinal diseases. NMN is being explored as a potential neuroprotective agent to help preserve or restore photoreceptor function after injury or degeneration.
These conditions share common features: oxidative stress, energy metabolism failure, and eventual photoreceptor death. This commonality is why researchers are interested in whether boosting NAD+ might provide broad retinal neuroprotection.
NMN, NAD+ and the Retina: What Laboratory Studies Are Finding
Recent experimental evidence has begun clarifying how NAD+ and NMN might protect retinal tissue. A March 2024 review article titled “Nicotinamide mononucleotide, a potential future treatment in ocular diseases” published in Graefe’s Archive for Clinical and Experimental Ophthalmology synthesized much of this data.
Themes from Preclinical Research
Across various experimental ocular diseases, NMN treatment has shown consistent effects:
Reduced oxidative stress in retinal cells
Decreased inflammatory signaling
Lower rates of cell death in photoreceptors and RPE
Improved energy metabolism markers
These findings suggest that NMN addresses fundamental mechanisms of retinal injury rather than just one specific pathway.
How NAD+ Supports Retinal Cells
The relationship between NAD+ and retinal health involves several interconnected mechanisms:
Mitochondrial Energy Production: Photoreceptor cells and retinal pigment epithelium cells have extraordinarily high energy demands. NAD+ is essential for mitochondrial respiration and ATP production. When NAD+ is adequate, mitochondrial function can be maintained even under stress.
Reactive Oxygen Species Management: NAD+ influences the activity of antioxidant systems that clear damaging ROS. In NAD+-deficient conditions, ROS accumulate and damage cellular components including DNA, proteins, and lipids.
Inflammatory Pathway Regulation: NAD+-dependent enzymes modulate inflammatory immune mediators including cytokine responses. Excessive inflammation damages retinal neurons and vasculature.
The Role of Sirtuins
NAD+ serves as a required substrate for sirtuins—a family of enzymes with the designation SIRT1 through SIRT7. Three sirtuins have received particular attention in retinal research:
| Sirtuin | Primary Retinal Functions |
|---|---|
| SIRT1 | DNA repair, stress response, photoreceptor protection |
| SIRT3 | Mitochondrial function, metabolic adaptation |
| SIRT6 | DNA repair, inflammation regulation |
| When NAD+ declines, sirtuin activity drops because these enzymes literally cannot function without their NAD+ cofactor. This is sometimes described as a metabolic sensor being deactivated—the cell loses its ability to detect and respond to damage. |
Research has shown that SIRT3 activity is impaired in NAD+-deficient retinas, reducing the retina’s capacity to adapt to metabolic stress. SIRT1 activation has been linked to reduced photoreceptor cell death under oxidative stress conditions.
Comparing NAD+ Precursors
Several NAD+ precursors have been tested in retinal research:
Nicotinamide (vitamin B3)
Nicotinamide mononucleotide (NMN)
Nicotinamide riboside (NR)
These compounds have overlapping but not identical effects. NMN is of particular interest because it enters the NAD+ biosynthesis pathway at a later stage, potentially offering more direct conversion. However, head-to-head comparisons in retinal disease models are limited.
Limitations to Keep in Mind
These mechanistic findings come primarily from rodent experiments and in vitro cell cultures. How closely they translate to human eyes remains an open question. Mouse retinas differ from human retinas in important ways, and conditions created by chemical stressors or genetic modifications don’t perfectly replicate the slow progression of human age-related disease.

Photoreceptor Degeneration in Retinal Detachment
Evidence for NMN in Retinal Detachment and Photoreceptor Degeneration
One of the most detailed examinations of NMN in a photoreceptor degenerative model comes from research using experimental retinal detachment in mice. Oxidative stress and inflammation are significant contributors to the development and progression of retinal diseases, including retinal detachment, ischemic retinopathy, glaucoma, and corneal defect. NMN’s effects are being studied in various models of ocular disease, such as corneal defect and dry eye.
In the experimental setup, tumor necrosis factor alpha, an inflammatory mediator, is implicated in photoreceptor cell death in retinal detachment models.
NMN administration after retinal detachment resulted in a significant reduction of TUNEL+ photoreceptors and CD11b+ macrophages, indicating its protective effect against retinal cell death and inflammation.
Importantly, delayed NMN administration still exerted protective effects on photoreceptors after retinal detachment.
NMN and Dry Eye
Research also shows that NMN has been shown to reduce inflammation and increase oil secretion in mouse models, helping treat dry eye conditions. NMN may alleviate dry eye symptoms by reducing inflammation and protecting corneal epithelial cells from hyperosmotic stress.
NMN and Cataracts
Additionally, NMN can inhibit the development of fibrotic cataracts by suppressing oxidative stress and cellular transitions leading to lens scarring.
Mechanistic insights reveal that oxidative stress leads to the production of reactive oxygen species (ROS), which can cause retinal cell death.
The Experimental Setup
Researchers induced retinal detachment in C57BL/6 mice aged 7-10 weeks by injecting sodium hyaluronate into the subretinal space. This mechanical separation triggers rapid photoreceptor death, providing a model to test potential protective interventions.
NMN was administered intraperitoneally (by injection into the abdominal cavity) at doses of 250-500 mg/kg/day. Researchers then measured various outcomes including photoreceptor survival, inflammatory markers, and oxidative damage.
Key Findings from the Retinal Detachment Model
The results demonstrated that NMN ameliorates photoreceptor cell death in this acute injury model:
Reduced Cell Death: NMN administration decreased TUNEL-positive cells (a marker of dying cells) in the early phase after retinal detachment (24 hours to 3 days). At the higher 500 mg/kg dose, the reduction was substantial.
Structural Protection: At 500 mg/kg, NMN preserved outer nuclear layer (ONL) thickness up to 7 days post-detachment. The ONL contains photoreceptor cell bodies, so preserving its thickness indicates that more photoreceptors survived.
Anti-inflammatory Effects: NMN decreased infiltration of CD11b+ macrophages into the subretinal space. These immune cells contribute to inflammation caused by the detachment. Reduced glial activation—measured by GFAP labeled glial activation—was also observed in early phases.
Antioxidant Protection: NMN normalized protein carbonyl content, a marker of oxidative damage. It also increased expression of heme oxygenase-1 (HO-1), an antioxidant enzyme.
Mechanistic Insights
The study provided evidence for how NMN exerts these effects:
NMN increased retinal NAD+ levels directly
SIRT1 protein expression increased in NMN-treated retinas
In 661W photoreceptor-like cells (a laboratory cell line), NMN improved survival under oxidative stress conditions
Experiments manipulating SIRT1 activity suggested that SIRT1 activation and downstream HO-1 induction significantly contribute to the protective effect
Notable Finding: Even delayed NMN treatment—starting 3 days after retinal detachment rather than immediately—conferred some structural protection and reduced macrophage infiltration. This suggests a potential therapeutic window beyond the immediate injury phase.
Translation Challenges
Before drawing conclusions about human use, several critical differences between this experiment and human supplementation must be acknowledged:
| Experimental Feature | Human Supplement Reality |
|---|---|
| Intraperitoneal injection | Oral ingestion |
| 250-500 mg/kg/day | Typical dose 250-500 mg total |
| Young, healthy mice | Older adults with comorbidities |
| Acute mechanical injury | Chronic degenerative disease |
| Controlled conditions | Variable real-world factors |
| A 250 mg/kg dose in a mouse would translate to an enormous dose in humans—one that nobody is taking as an oral supplement. Additionally, injecting a compound directly into the abdominal cavity achieves different tissue levels than swallowing a capsule. |
This research demonstrates that NMN can protect photoreceptors in an experimental setting. It does not establish that oral NMN supplements at typical human doses will protect against age-related vision loss.
NMN and Age-Related Macular Degeneration, Diabetic Retinopathy, and Other Retinal Diseases
Beyond acute injury models, researchers have examined NAD+ boosters in the context of chronic retinal diseases. A review funded by the National Research Foundation of Korea (grant RS-2021-NR060133) titled “Preventive and Protective Effects of Nicotinamide Adenine Dinucleotide Boosters in Aging and Retinal Diseases” synthesized much of this work.
NAD+ Boosters in AMD Models
Research using models that mimic aspects of age related macular degeneration has produced encouraging results. Studies from Tongji University published in Oxidative Medicine and Cellular Longevity exposed human retinal cells to sodium iodate (NaIO₃), a chemical stressor that induces retinal degeneration resembling AMD.
Key findings from this research:
NMN treatment resulted in a 50% reduction in DNA damage in stressed human retinal cells
The percentage of senescent cells dropped following NMN treatment
Reactive oxygen species levels were suppressed
In the mouse model component of this research (using C57BL/6J male mice injected with NaIO₃ and treated with NMN at 300 mg/kg intraperitoneally):
Structural changes in mouse retinas were mitigated
Senescent cell propagation was prevented
NAD+ levels in retinal tissue were restored
The researchers proposed that NMN works by boosting NAD+, which then fuels enzymes capable of repairing DNA and reducing ROS. This goes beyond a simple “antioxidant” explanation to identify specific pathways involved.
The Macrophage Connection
A separate line of research (Apte and colleagues) revealed that NMN’s benefits in AMD-like conditions may work partly through immune cells, not just direct photoreceptor protection.
The study found that age-associated senescence in macrophages—immune cells that accumulate in the eye with AMD—was reversed by NMN treatment. Specifically:
Inhibiting NAD+ biosynthesis in healthy mouse macrophages increased expression of senescence-associated genes p16 and p21
NMN treatment suppressed senescence markers in macrophages from an AMD model
In AMD model mice, NMN injections suppressed subretinal drusenoid deposits (SDDs), pathological hallmarks of dry AMD
Electroretinography showed that NMN improved photoreceptor responses to light, indicating functional improvement in retinal function
Convergent Evidence: Researchers also tested dasatinib and quercetin—drugs that selectively eliminate senescent cells—and found similar benefits. This convergence strengthens the evidence that senescent cell accumulation is mechanistically important in AMD pathology, and that NMN eliminates senescent cells or reverses their dysfunction.
Diabetic Retinopathy Research
In preclinical models of diabetic retinopathy, NAD+ boosters have shown potential to:
Preserve retinal energy metabolism
Protect retinal ganglion cells from degeneration
Reduce gliosis (scarring by glial cells)
Mitigate microvascular injury
However, NMN’s benefits in diabetic models may be partly indirect. Improving systemic metabolism and glycemic control—documented effects of NAD+ boosting in animal studies—could benefit the retina through better overall metabolic health rather than through direct retinal action alone.
This distinction matters because managing ocular diseases requires addressing underlying systemic conditions. If NMN helps diabetic retinopathy partly by improving metabolic control, that’s valuable but different from direct retinal neuroprotection.
Other Conditions Under Investigation
NAD+ boosters are being explored experimentally in several other ocular diseases:
Ischemic retinopathy: Conditions involving inadequate blood flow to the retina
Light-induced retinal damage: Excessive light exposure causing photoreceptor injury
Glaucomatous optic neuropathy: Disease affecting retinal ganglion cells and the optic nerve
Most of this research focuses on sirtuin pathways and their role in protecting neurons under stress. The evidence base is less developed than for AMD and diabetic retinopathy but follows similar mechanistic logic.
The Current State of Human Evidence
Here is the critical point that must be emphasized: as of mid-2026, robust human clinical trials specifically testing NMN for AMD, diabetic retinopathy, or other retinal diseases are limited or absent.
The discussion above is based on:
Mechanistic plausibility
Cell culture experiments
Animal model data
We do not have randomized controlled trials showing that people who take NMN supplements experience slower AMD progression, better visual acuity, or reduced diabetic retinopathy severity compared to those taking placebo.
This gap between preclinical stages and clinical evidence is where NMN currently sits in eye health research.
Safety, Open Questions, and Practical Considerations
Human Studies on NMN Safety
Given the limited human evidence for eye-specific benefits, what do we know about NMN safety and what questions remain?
Available human studies of oral NMN—typically at doses ranging from 250-900 mg/day over periods of several weeks to months—have generally reported good short-term tolerability. Blood levels of NAD+ metabolites increase, indicating the supplement is absorbed and metabolized.
However, these studies were designed to assess general safety and NAD+ elevation, not eye-specific outcomes. They do not tell us:
Whether systemic NAD+ elevation translates to increased NAD+ in retinal tissue
What the long-term effects of sustained NAD+ elevation might be
How NMN interacts with medications commonly used by older adults
Whether people with existing eye disease respond differently than healthy individuals
Complexity of NAD+ Biology
NAD+ metabolism is not a simple “fill the tank” situation. The biology involves multiple competing pathways, and manipulating one can affect others.
One concern raised in research literature: excessive NMN or high NMN/NAD+ ratios may activate pathways like SARM1 in some experimental models. SARM1 is implicated in pathologic signaling pathways that can trigger axon degeneration. While this has been demonstrated in specific experimental contexts (particularly related to nerve injury), the clinical relevance for typical human NMN supplement use is not understood.
This observation underscores that more is not necessarily better. Evidence-based dosing—when we eventually have the evidence—will be crucial.
Key Knowledge Gaps
Several fundamental questions remain unanswered:
Tissue penetration: Does systemically elevated NAD+ reliably increase NAD+ levels in human retinal tissue? The retina sits behind the blood-retinal barrier, which may limit access.
Dose-response: What dose (if any) produces eye-related benefits? Animal studies use doses not directly comparable to human supplements.
Patient selection: Would early AMD patients respond differently than those with advanced disease? Would non-proliferative diabetic retinopathy patients see different effects than those with proliferative disease?
Outcome measures: What endpoints should clinical trials measure—visual acuity, OCT-measured retinal thickness, progression rates, something else?
Practical Guidance
Based on current evidence, here is a non-prescriptive framework for thinking about NMN and eye health:
NMN should not replace proven therapies. If you have wet AMD, anti-VEGF injections remain the standard of care. If you have diabetic retinopathy, glycemic control, blood pressure management, and appropriate ophthalmic treatment are essential. NMN is not an alternative to these interventions.
Discuss supplements with your healthcare team. If you’re considering NMN, mention it to your ophthalmologist and primary care physician, particularly if you have glaucoma, AMD, diabetic retinopathy, or take medications like anticoagulants.
View NMN as experimental for eye health. The therapeutic potential exists based on mechanism, but clinical trials haven’t confirmed benefits for vision.
Questions to Ask Your Doctor:
Given my eye condition, are there any concerns about taking NMN supplements?
What proven interventions should I prioritize for my specific situation?
Are there clinical trials for NMN or similar compounds that I might be eligible for?
How often should I have my eyes examined given my risk factors?
Beyond NMN: Evidence-Based Strategies to Support Eye Health
While NMN research continues, several strategies already have stronger evidence for reducing risk or slowing progression of macular degeneration and other eye diseases.
AREDS and AREDS2 Formulations
The Age-Related Eye Disease Studies established that specific supplement combinations can benefit certain AMD patients:
AREDS2 Formula Components:
Vitamin C (500 mg)
Vitamin E (400 IU)
Zinc (80 mg as zinc oxide)
Copper (2 mg as cupric oxide)
Lutein (10 mg)
Zeaxanthin (2 mg)
These formulations can reduce the risk of progression from intermediate to advanced AMD in appropriate patients. Important clarifications:
These are not general multivitamins—they’re high-dose formulations
They’re intended for people with intermediate AMD, not for prevention in people with no disease
They should be used under professional guidance
Lifestyle Measures with Strong Evidence
Smoking cessation: One of the strongest modifiable risk factors for AMD. If you smoke and are concerned about your vision, quitting is likely more impactful than any supplement.
Cardiovascular health: Maintaining healthy blood pressure, blood sugar, and cholesterol supports the tiny blood vessels that nourish your retina. The same factors that protect your heart protect your eyes.
Physical activity and weight management: May support healthy NAD+ metabolism naturally, in addition to their cardiovascular benefits.
Dietary Patterns for Eye Health
Research consistently links certain dietary patterns with better eye health outcomes:
| Food Category | Examples | Key Nutrients |
|---|---|---|
| Dark leafy greens | Spinach, kale, collards | Lutein, zeaxanthin |
| Colorful vegetables | Carrots, bell peppers, tomatoes | Carotenoids, vitamin C |
| Fatty fish | Salmon, mackerel, sardines | Omega-3 fatty acids |
| Nuts and seeds | Almonds, walnuts, flaxseed | Vitamin E, healthy fats |
| Whole grains | Oats, quinoa, brown rice | B vitamins, zinc |
 |
Many of these foods contain compounds—polyphenols, carotenoids, omega-3 fatty acids—that may interact favorably with NAD+ pathways and antioxidant defenses. Eating for eye health isn’t separate from eating for overall health.
Environmental and Behavioral Habits
UV protection: Use sunglasses blocking 99-100% of UVA and UVB rays when outdoors
Screen habits: Take regular breaks during prolonged screen use; good lighting reduces strain
Regular eye exams: Dilated exams annually after age 60, or earlier and more frequently for people with diabetes or strong family history of eye disease
Putting NMN in Context
If you choose to consider NMN, view it as a possible adjunct to—not a replacement for—these established protective strategies. The foundation of eye health remains:
Regular professional monitoring
Management of systemic conditions
Proven lifestyle interventions
Evidence-based supplements where indicated
Potential future NMN treatment protocols, if they emerge from clinical trials, would add to this foundation rather than replace it.
How Might Future Research Clarify NMN’s Role in Protecting Vision?
The gap between promising laboratory findings and clinical application creates an urgent need for further research. What kinds of studies would help clarify whether NMN belongs in an eye health strategy?
Types of Studies Needed
Randomized controlled trials remain the gold standard. We need studies that:
Recruit people with early or intermediate AMD and randomly assign them to NMN or placebo
Measure meaningful outcomes: visual acuity, OCT-based retinal structure, disease progression rates
Run long enough to detect differences (likely 1-3 years minimum for slowly progressive conditions)
For diabetic retinopathy, trials examining whether NMN affects microvascular changes, neurodegeneration markers, or progression to more severe stages would be valuable.
Dose-Finding and Duration Questions
Current animal studies use doses not directly applicable to human supplementation. We need:
Studies determining the minimal effective dose (if any) for eye-related outcomes
Comparisons between NMN, nicotinamide riboside, and nicotinamide to identify whether one is superior
Long-term safety data, particularly in older populations already taking multiple medications
Biomarker Development
A practical challenge: we can’t easily measure NAD+ levels in living human retinas. Research priorities include:
Developing markers of retinal NAD+ status, sirtuin activity, or oxidative damage measurable through blood tests or imaging
Using advanced techniques like OCT-angiography or adaptive optics imaging to detect subtle structural changes that might indicate NAD+ boosting benefits
Correlating systemic NAD+ metabolite levels with retinal health outcomes
Open Questions Worth Investigating
Several thought-provoking questions deserve investigation:
Genetic variation: Could people with certain genetic backgrounds—perhaps variants in complement genes or mitochondrial genes associated with AMD risk—respond differently to NMN?
Local delivery: Might eye drops or intravitreal injections of NAD+-related compounds be more effective than oral supplementation for reaching retinal tissue?
Combination approaches: How could NMN be safely combined with established treatments like anti-VEGF injections or AREDS2 supplements? Could combinations improve outcomes or reduce risks?
Mechanism specificity: Does NMN work primarily by reversing senescence, enhancing mitochondrial function, reducing inflammation caused by various factors, or through multiple pathways simultaneously?
Staying Informed
If you’re interested in this topic, follow developments in peer-reviewed ophthalmology and aging research rather than relying solely on supplement marketing claims. Resources include:
PubMed searches for “NMN retina” or “NAD+ macular degeneration”
Major ophthalmology journals like JAMA Ophthalmology, Ophthalmology, and Investigative Ophthalmology & Visual Science
Clinical trial registries like ClinicalTrials.gov
When you encounter new research, bring it to your clinician for discussion. They can help you evaluate whether findings are relevant to your situation.
Conclusion: Where Does NMN Fit in an Eye-Health Strategy Today?
Nicotinamide mononucleotide is a promising NAD+ precursor that has demonstrated neuroprotective effects in animal and cell models of retinal disease. However, its role in preventing macular degeneration and vision loss in humans remains unproven.
Here’s what we can say with confidence:
The retina is metabolically demanding and sensitive to NAD+ availability and mitochondrial function
NMN has demonstrated protective effects against ocular inflammation, oxidative stress, and photoreceptor degeneration in preclinical models, including retinal detachment and AMD-like conditions
Evidence-based eye care currently relies on established interventions: AREDS2 supplements where appropriate, lifestyle modifications, systemic disease control, and standard ophthalmic treatments for conditions like wet AMD and diabetic retinopathy
If you’re concerned about your vision—particularly if you have risk factors for macular degeneration or diabetic eye disease—your foundation should be:
Regular dilated eye exams with qualified professionals
Medical management of conditions like diabetes, hypertension, and cardiovascular disease
Lifestyle measures with documented benefits (not smoking, healthy diet, UV protection)
If you choose to explore NMN supplementation, view it as an experimental adjunct and discuss it openly with your eye-care and primary-care clinicians. They can help you weigh potential benefits against unknowns and ensure any supplement doesn’t interfere with your other treatments.
The urge future research will address is whether NMN’s promising laboratory signals translate into clinical benefit. Until that research is completed, we’re in a position of informed uncertainty—knowing enough to be interested, but not enough to make definitive recommendations.
Stay curious, stay informed, and keep having those conversations with your healthcare team about what’s working and what’s worth trying.
The Role of Oxidative Stress in Retinal Aging and Disease
Oxidative stress is a central player in the aging of the retina and the development of major ocular diseases, including age related macular degeneration (AMD). The retina, and especially the retinal pigment epithelial cells (RPE), are constantly exposed to high levels of metabolic activity and light, making them particularly susceptible to the damaging effects of reactive oxygen species. Over time, this oxidative burden can overwhelm the retina’s natural defenses, leading to cellular senescence in RPE cells. These senescent cells not only lose their ability to support photoreceptor health but also begin to secrete inflammatory immune mediators and dysregulated cytokine responses, fueling ocular inflammation and accelerating photoreceptor death.
A key factor in this process is the decline of nicotinamide adenine dinucleotide (NAD+) with age. NAD+ is essential for mitochondrial function and cellular energy metabolism. As NAD+ levels drop, mitochondrial dysfunction and complex metabolic dysregulation set in, making retinal cells more vulnerable to oxidative stress and triggering a cascade of events that culminate in retinal degeneration. This is particularly evident in experimental ocular diseases such as retinal detachment, diabetic retinopathy, and light induced retinal degeneration, where oxidative stress is a hallmark feature and a major driver of photoreceptor cell death.
In these photoreceptor degenerative models, research has shown that nicotinamide mononucleotide (NMN) treatment can significantly ameliorate photoreceptor cell death and improve retinal function. NMN acts by replenishing NAD+ levels, which helps restore mitochondrial function, reduce oxidative stress, and modulate pathologic signaling pathways involved in inflammation and cell death. Notably, NMN’s neuroprotective effects extend to preventing apoptotic photoreceptor degeneration and reducing axon degeneration that can be triggered by imbalances in NMN/NAD+ ratios.
Managing ocular diseases requires a multifaceted approach that addresses not only the symptoms but also the underlying mechanisms—such as oxidative stress, inflammation, and metabolic dysfunction. NMN treatment has demonstrated the ability to reduce these damaging processes in various experimental ocular diseases, supporting its therapeutic potential for treating chronic conditions involving retinal neurodegeneration, including diabetic retinopathy and retinal detachment. While clinical evidence is still emerging, preclinical studies provide a strong rationale for further investigation into NMN and other NAD+ precursors as strategies to protect against retinal degeneration and promote retinal health.
Ultimately, the interplay between oxidative stress, cellular senescence, and declining NAD+ metabolism forms a complex web that underlies many forms of retinal disease. By targeting these interconnected pathways—through interventions like NMN that improve mitochondrial function, reduce inflammation, and support cellular resilience—we may be able to develop more effective therapies for preventing and treating age related macular degeneration and other ocular diseases. Continued research will be essential to translate these promising findings from the laboratory into clinical practice, offering hope for improved management of retinal aging and vision loss.
NMN Supplementation: Forms, Dosing, and Bioavailability
As interest in nicotinamide mononucleotide (NMN) grows for its potential to support eye health and combat age related macular degeneration, understanding how NMN supplements are formulated, dosed, and absorbed becomes increasingly important. Since NMN acts as a direct precursor to nicotinamide adenine dinucleotide (NAD+), optimizing its delivery could be key to harnessing its benefits for ocular diseases such as diabetic retinopathy and other conditions linked to impaired energy metabolism.
Further Reading
Explore more articles related to this topic:
- NMN and Brain Health: Can Boosting NAD+ Prevent Cognitive Decline?
- Health Benefits of NMN: What Science Really Shows
- NMN Research Update 2026: What Recent Human Trials Tell Us About Reversing Biological Age
- NMN for Skin Aging: Can Boosting NAD+ Actually Reduce Wrinkles and Improve Elasticity?
- Best Longevity Supplements in 2026: Science-Backed Picks Only