The question of whether nicotinamide mononucleotide poses risks to your liver has become increasingly important as more people explore this naturally occurring molecule for its potential benefits. With conflicting information online and limited long-term data, understanding what the science actually shows—and what it doesn’t—is essential before making any decisions about supplementation. Research indicates that decreased NAD+ levels with aging are associated with various health conditions, including Alzheimer’s disease, Parkinson’s disease, type 2 diabetes, muscular atrophy, and cardiovascular conditions.
This comprehensive guide examines the current evidence on NMN and liver safety, breaking down human clinical trials, animal studies, and the practical considerations you need to know.
Introduction to Nicotinamide Mononucleotide
Nicotinamide mononucleotide (NMN) is a naturally occurring molecule that has rapidly gained attention in the world of health and longevity research. As a direct precursor to nicotinamide adenine dinucleotide (NAD+), NMN plays an essential role in supporting healthy cells, facilitating DNA repair, and powering the energy production processes that keep our bodies functioning optimally. With age, NAD+ levels naturally decline, which has prompted scientists to explore whether NMN supplements could help restore these levels and promote better health outcomes.
Recent NMN research has focused on the potential benefits of NMN supplementation, particularly its effects on insulin sensitivity, muscle function, and aerobic capacity. Animal studies have shown that NMN can improve health effects in aged mice, including increased insulin sensitivity and enhanced physical performance. These promising results have led to a surge in clinical trials involving humans, where NMN has been tested in various populations, such as postmenopausal women and healthy Japanese men. Verification successful studies have demonstrated that NMN supplementation can lead to increased insulin sensitivity and improved metabolic markers, with some evidence suggesting that higher doses may be more effective than medium doses.
Despite these encouraging findings, it is important to recognize that NMN research is still in its early stages. Most human studies to date have been small, short-term, and often limited to healthy individuals. While double-blind, placebo-controlled trials have begun to confirm the efficacy and safety of NMN supplements in certain groups, more studies are needed to fully understand the long-term effects, optimal dosing, and potential risks. For example, while NMN has shown benefits in animal models, questions remain about its impact on organs such as the kidneys and its theoretical links to cancer risk.
Nicotinamide riboside (NR), another NAD+ precursor, has also been studied for similar potential benefits, but direct comparisons between NR and NMN are still limited. As the body of evidence grows, it will be essential to conduct more rigorous clinical trials to confirm the efficacy and safety of NMN and to clarify how it compares to other supplements.
In summary, NMN is an exciting area of research with the potential to support longevity and overall health. However, as with any supplement, it is essential to approach NMN supplementation with caution. Consulting a healthcare professional before starting NMN—especially for those with underlying health conditions or who are taking other medications—is strongly recommended. Continued research and careful verification of NMN supplements will be crucial to unlocking their full potential and ensuring their safe use for supporting healthy aging.
Quick Answer: What Do We Know About NMN and Liver Safety in 2024?
Based on available data through 2023-2024, NMN has not shown clear signs of liver toxicity in healthy human adults during short-term use. Studies administering doses up to 1,200–1,250 mg/day for periods of 4–12 weeks have not reported significant elevations in liver enzymes or serious liver-related adverse events. Participants in these clinical trials maintained normal ranges for key markers like ALT, AST, and bilirubin throughout the study periods.
However, this evidence comes with important caveats. Most human research involves small sample sizes (often fewer than 50 participants), limited durations, and generally healthy populations without pre-existing liver conditions. Long-term liver safety—spanning years of continuous use—remains essentially unknown. Similarly, how NMN might affect people with fatty liver disease, hepatitis, cirrhosis, or those taking multiple medications has not been systematically studied.
On the potentially positive side, some animal studies and mechanistic research suggest NMN may actually support liver function. Data from aged mice shows improved markers of liver health, reduced fat accumulation, and better insulin sensitivity. These findings are intriguing but cannot be directly assumed to translate to human outcomes.
Important note: This article is informational only and does not constitute medical advice. Anyone with existing liver conditions, those taking medications that affect the liver, or individuals with other health concerns should discuss NMN with a qualified healthcare professional before use.
| Current Evidence Summary | Status |
|---|---|
| Short-term use in healthy adults | Generally well tolerated |
| Long-term continuous use | Unknown |
| People with liver disease | Consult your doctor first |
| Animal model data | Suggests potential protective effects |
What Is NMN and How Is It Linked to the Liver?
Nicotinamide mononucleotide is a compound your body naturally produces as part of vitamin B3 metabolism. It serves as a direct precursor to nicotinamide adenine dinucleotide (NAD+), a coenzyme found in every cell of your body. Think of NMN as the raw material your cells use to manufacture NAD+, which then powers hundreds of essential biological processes.
NAD+ plays a particularly critical role in liver cells (hepatocytes). Your liver is one of the most metabolically active organs in your body, responsible for detoxification, protein synthesis, bile production, and energy metabolism. NAD+ is essential for these functions—it enables fatty acid oxidation, supports DNA repair mechanisms, and powers the mitochondria that generate cellular energy. Without adequate NAD+, liver cells struggle to perform their basic functions effectively.
Research has shown that NAD+ levels tend to decline with age and in various metabolic conditions. People with non-alcoholic fatty liver disease, type 2 diabetes, and obesity often show reduced hepatic NAD+ levels. This decline has made NMN a focus of longevity and metabolic research, with scientists investigating whether restoring NAD+ through supplementation might improve age related diseases and metabolic dysfunction.
It’s worth noting that in 2022, the U.S. FDA determined that NMN could not be lawfully marketed as a dietary supplement because it had been investigated as a drug. This regulatory decision affects availability and how products can be labeled in the United States, but it does not necessarily reflect safety concerns about the compound itself.
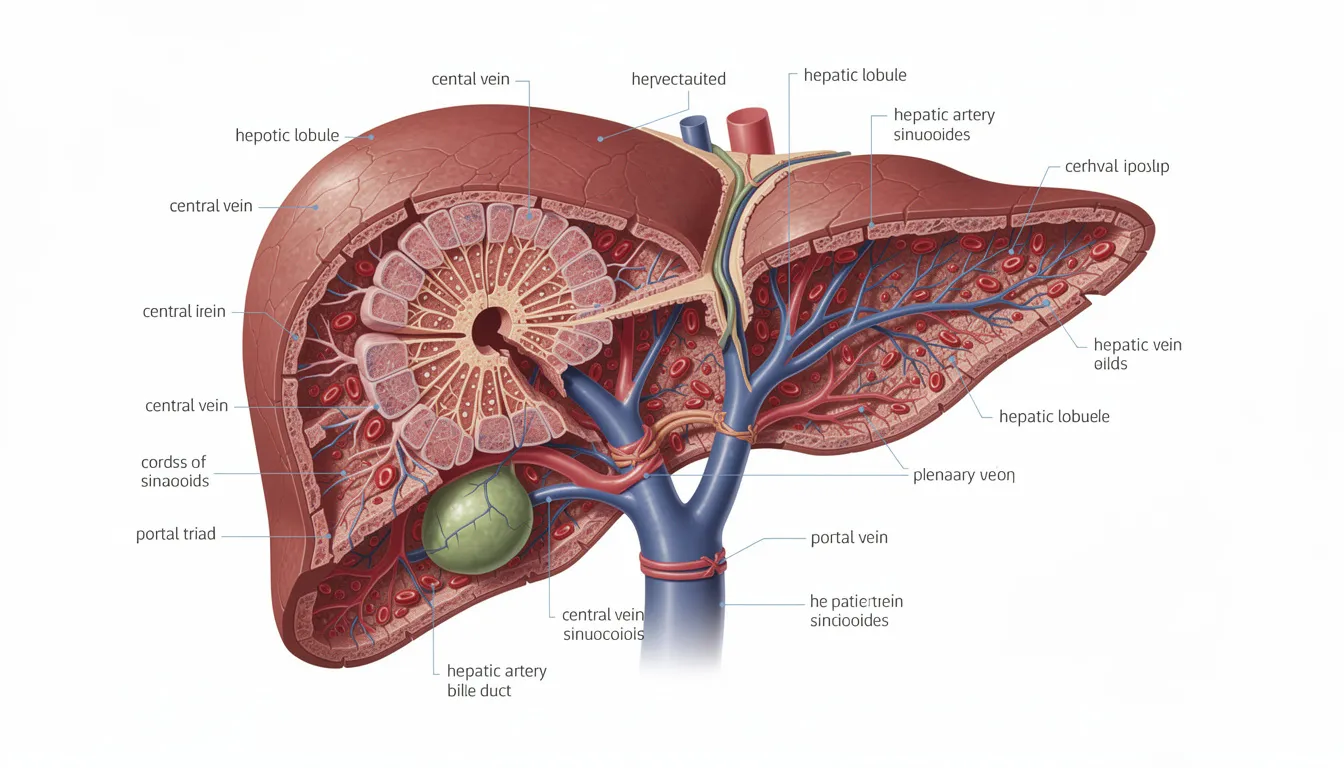
How the Liver Metabolizes NMN and NAD⁺: Pathways and Mechanisms
The liver serves as a central hub for processing vitamin B3-related compounds, including NMN, niacin, and nicotinamide. Understanding how your liver handles these molecules helps explain why researchers pay close attention to hepatic safety when studying NMN supplementation.
When you take NMN through oral administration, the absorption pathway is more complex than simply having the molecule travel intact to your liver. Current evidence suggests that oral NMN may be partially converted to nicotinamide riboside (NR) or nicotinamide (NAM) during absorption in the gut. These compounds then enter circulation, get taken up by liver cells, and are reconverted back to NMN and ultimately to NAD+ through what’s called the salvage pathway.
The NAD+ salvage pathway in the liver works like this:
Nicotinamide (NAM) is converted to NMN by an enzyme called NAMPT
NMN is then converted to NAD+ by NMNAT enzymes
NAD+ is used by cells and eventually broken back down to nicotinamide
The cycle repeats
This pathway is the dominant route for maintaining NAD+ levels in adult liver tissue. Research shows that beef liver tissue exhibits exceptionally high NMN catabolizing activity—about 16 times higher than other enzymatic processes in the liver—indicating that hepatic NMN metabolism is highly active.
Once NAD+ is utilized and broken down, the resulting nicotinamide metabolite levels include compounds like N-methyl-nicotinamide (MNA), 2-pyridone (2-PY), and 4-pyridone (4-PY). These metabolites are cleared primarily through the kidneys. Because the liver is central to these conversions, any large, chronic increase in NAD+ turnover could theoretically stress hepatic and renal excretion pathways. This is precisely why safety research carefully monitors liver enzymes and downstream metabolites.

Oral Administration and Bioavailability: How NMN Reaches the Liver
When it comes to NMN supplements, how the compound is delivered and absorbed by the body is a key factor in determining its potential benefits. Oral administration is the most common method, and research shows that NMN is efficiently absorbed through the digestive tract and enters the bloodstream. From there, it travels directly to the liver via the hepatic portal vein—a critical step, since the liver acts as a central processing hub for many nutrients and supplements.
Once NMN reaches the liver, it is rapidly converted into nicotinamide adenine dinucleotide (NAD+), a naturally occurring molecule that is essential for cellular energy production, DNA repair, and overall metabolic health. By boosting NAD+ levels in the liver, NMN supplementation may help support vital processes such as improved insulin sensitivity and enhanced aerobic capacity, both of which are important for maintaining metabolic health and reducing the risk of age related diseases.
Several studies have demonstrated that oral administration of NMN can increase NAD+ concentrations in the liver, which may translate into real-world health benefits. For example, increased NAD+ supports more efficient DNA repair and cellular resilience, potentially contributing to healthier aging. Improved insulin sensitivity, observed in some clinical trials, suggests that NMN could play a role in metabolic regulation and disease prevention. In clinical and animal studies, medium dose NMN supplementation (such as 600 mg or 300 mg twice daily) has been compared with higher or lower doses to evaluate safety, efficacy, and benefits related to NAD+ levels, metabolic health, and exercise capacity.
However, it’s important to note that while these findings are promising, more studies are needed to fully understand the bioavailability and pharmacokinetics of NMN supplements in humans. Factors such as dosage, formulation, and individual differences can all affect how much NMN actually reaches the liver and how effectively it is converted to NAD+. As NMN research continues, future studies will help clarify the efficacy of oral administration and guide best practices for supplement use.
Common dosages for NMN supplements range from 250 milligrams to 1,000 milligrams per day, with a suggested daily dosage of 500 milligrams considered effective and safe for most users. The NMN content in commercially available supplements typically ranges from 50 to 150 milligrams per capsule.
What Human Studies Say About NMN and Liver Function
Human clinical trials investigating NMN remain relatively limited in scope. The studies published to date typically involve tens of participants (not hundreds or thousands), run for 4–12 weeks, and generally enroll adults without serious pre-existing liver disease. Despite these limitations, the available data provides a foundation for understanding NMN’s short-term liver safety profile.
For example, a double blind study conducted in Japan tested high-dose NMN supplementation in healthy adults, emphasizing the rigorous design and reliability of the results. In another study involving postmenopausal women with prediabetes, NMN administration was shown to improve insulin sensitivity and glucose metabolism in overweight or obese individuals with prediabetes.
Overview of Key Clinical Trials
Japanese Safety Studies (2016–2020)
Early human research on NMN focused primarily on establishing basic safety and tolerability. Studies in healthy Japanese men and other adult populations examined both single-dose and repeated-dose protocols up to approximately 500 mg/day. These trials reported no clinically meaningful changes in liver enzymes—including ALT, AST, ALP, and GGT—with values remaining within reference ranges throughout the study periods.
Postmenopausal Women with Prediabetes (2021)
A randomized, placebo controlled trial examined 250 mg/day of NMN for 10 weeks in prediabetic women. The primary aim was to assess effects on muscle insulin sensitivity and metabolic parameters. From a liver safety perspective, the study reported no significant NMN-related liver enzyme elevations compared with the placebo group. This trial provided evidence that NMN was well tolerated in a population with underlying metabolic dysfunction.
High-Dose Japanese Trial (2022–2023)
Perhaps the most relevant study for liver safety questions is a double blind trial that administered 1,250 mg/day of β-NMN for 4 weeks to 31 healthy adults aged 20–65. This represents a high dose compared to many other studies.
The study design included:
Randomized, double blind, placebo controlled, parallel-group structure
Assessments of hematology, clinical biochemistry (including liver enzymes), urinalysis, vital signs, and adverse events
Measurements at baseline, week 2, week 4, and follow-up
The main finding relevant to liver safety: liver-related markers stayed within normal ranges and did not differ significantly from placebo. Verification successful of these clinical parameters suggested the high dose was tolerated without obvious hepatic stress.
| Trial | Year | Population | Dose | Duration | Liver Outcomes |
|---|---|---|---|---|---|
| Japanese safety | 2016-2020 | Healthy adults | Up to 500 mg/day | Varied | No enzyme changes |
| Prediabetes study | 2021 | Postmenopausal women | 250 mg/day | 10 weeks | No elevations vs placebo |
| High-dose trial | 2022-2023 | Healthy adults | 1,250 mg/day | 4 weeks | Normal ranges maintained |
Monitoring Liver Markers in NMN Trials
Clinical researchers use a standard panel of blood tests to monitor liver safety in supplement trials. These include:
ALT (alanine aminotransferase): Elevated when liver cells are damaged
AST (aspartate aminotransferase): Another marker of liver cell injury
ALP (alkaline phosphatase): Can indicate bile duct problems
GGT (gamma-glutamyl transferase): Sensitive to various liver conditions
Total bilirubin: Measures liver’s ability to process waste
Albumin: Reflects liver’s protein-producing function
In the 1,250 mg/day study, all these markers remained within clinical reference ranges and showed no NMN-specific patterns of change versus placebo. Some trials also track lipid profiles (triglycerides, LDL, HDL) and fasting glucose/insulin, which relate to metabolic liver health but are not direct toxicity markers.
It’s crucial to understand that “within normal range” does not equate to proven safety in all contexts. It only indicates no obvious liver injury under the specific conditions tested—healthy adults, limited duration, controlled environments. More studies are needed to confirm these findings across broader populations and longer timeframes.
Animal and Cell Studies: Potential Liver Benefits and Theoretical Risks
Much of what we know about NMN and liver function comes from mouse models and cell culture experiments conducted between approximately 2013 and 2023. While these studies cannot directly predict human outcomes, they provide mechanistic insights that help explain how NMN might affect liver biology.
Evidence Supporting Liver Protection
Research in aged mice has produced some compelling data. A 2024 study from Tsinghua University examined how NMN affected liver resilience to drug-induced injury. Researchers injected 96-week-old mice (roughly equivalent to 65 human years) with 500 mg/kg of NMN, then exposed them to acetaminophen (APAP)—a compound that causes severe liver toxicity and represents the leading cause of acute liver failure.
The results were notable:
Liver damage markers (ALT and AST blood levels) returned to youthful baseline levels in NMN-treated mice
Untreated aged mice showed significantly elevated markers indicating substantial hepatic damage
The protective effect appeared to operate through activation of SIRT3, a sirtuin enzyme that regulates mitochondrial function
The mechanism involves multiple pathways. NMN activates sirtuin enzymes, which use NAD+ as a cofactor. When researchers genetically removed SIRT3 from mice, many protective effects of NMN were abolished—demonstrating that SIRT3 is essential for these hepatoprotective benefits.
Additionally, NMN enhanced antioxidant defenses by promoting activation of Nrf2, one of the most important regulators of oxidative stress response in cells. In aged mice without NMN treatment, Nrf2 failed to relocate to the cell nucleus in response to liver injury. When liver-injured aged mice received NMN, Nrf2 successfully transferred to the nucleus where it could activate antioxidant genes.
Studies examining NMN’s role in alcohol-related liver disease have also shown promise. Research demonstrates that NMN supplementation can protect against some forms of alcohol-induced liver damage, though this protection depends on functional NMNAT1 enzymes being present to convert NMN to NAD+.
In several models of obesity-related non-alcoholic fatty liver disease, NMN improved markers like hepatic triglyceride content and inflammatory signaling. Previous work has also shown that NMN inhibits the onset of liver fibrosis in mice by improving telomere length—suggesting potential applications in chronic liver disease management.
Theoretical Concerns and Unresolved Questions
While the animal data often appears positive, some scientists have raised theoretical concerns about chronically elevating NAD+ levels:
Potential support of abnormal cells: NAD+ is involved in DNA repair and cell survival. Some researchers speculate that boosting NAD+ could theoretically support the survival of pre-existing abnormal or malignant cells. This is an active area of basic research, though no clinical trials have demonstrated that NMN causes cancer or liver tumors in humans.
Metabolite accumulation: Increasing NAD+ turnover produces downstream metabolites (MNA, 2-PY, 4-PY) that, at very high levels, have been associated with cardiovascular and kidney risk in some observational work. Whether these associations are causal and whether they apply to liver health specifically remains unclear.
Distinction from human data: Human studies have found that NMN reduces blood fat levels, which has been linked to fatty liver disease. However, one study found that NMN does not affect liver fat mass in humans—suggesting that while NMN may reduce circulating lipids, it may not directly reduce hepatic lipid accumulation.
These potential risks remain largely theoretical in the context of human NMN use and have not been clearly demonstrated as liver-specific toxicity in controlled studies.
Mouse Dose vs. Human Dose: Why Translation Is Tricky
A critical factor when interpreting animal studies is dose translation. Many rodent NMN experiments use doses like 300–500 mg/kg/day, which might sound comparable to human supplementation but actually aren’t.
A 300 mg/kg dose in a mouse does not equal 300 mg/kg in a human. When adjusted for body surface area and metabolic differences, the human-equivalent dose is much lower. For example:
| Mouse Dose | Approximate Human Equivalent* |
|---|---|
| 100 mg/kg | ~8 mg/kg (560 mg for 70kg person) |
| 300 mg/kg | ~24 mg/kg (1,680 mg for 70kg person) |
| 500 mg/kg | ~40 mg/kg (2,800 mg for 70kg person) |
| *Rough estimates using standard body surface area conversion |
Very high NAD+ precursor doses in animals can reveal potential toxicity thresholds that humans may never reach with common supplemental ranges (typically 250–1,000 mg/day). This makes direct extrapolation from animal toxicity studies to human safety assessments challenging.

Does NMN Cause Liver Toxicity? Current Evidence and Open Questions
Based on data available through 2024, NMN has not been clearly linked to liver toxicity in healthy human participants over periods spanning weeks to a few months. This is an important finding but comes with significant qualifications.
The common patterns seen with known hepatotoxic supplements—significant sustained ALT/AST elevation, jaundice, hospitalization for acute liver injury—have not been reported in published NMN trials. The evidence to date suggests that taking NMN at doses studied in research does not produce obvious signs of liver damage in healthy adults during short-term use.
Comparison with Related Compounds
It’s instructive to compare NMN’s emerging safety profile with other NAD+ precursors and related B vitamins:
Niacin (nicotinic acid): Effective for improving lipid profiles but well-known for causing flushing at therapeutic doses. More concerning, high-dose niacin has documented hepatotoxicity potential—liver strain and even failure have been reported with gram-level sustained-release formulations.
Nicotinamide (NAM): Generally considered safer than niacin, but can cause liver issues at multi-gram doses in some individuals. Liver enzyme elevations have been documented in clinical use at high doses.
NMN: So far appears distinct from both—no vasodilatory flushing and no clear hepatotoxic signal in short-term data.
Nicotinamide riboside: Has more published human data and is generally regarded as safe by FDA. No major liver safety signal to date.
Because all these molecules feed into the same NAD+ network, some safety considerations (such as metabolite buildup) may overlap. However, each has its own pharmacokinetics and side-effect profile. As of 2024, no large head-to-head trial has directly compared NMN and NR specifically on liver outcomes in humans.
| Compound | Liver Safety Profile | Known Concerns |
|---|---|---|
| NMN | No clear toxicity signal (limited data) | Long-term effects unknown |
| Nicotinamide riboside | Generally well tolerated | More human data available |
| Nicotinic acid (niacin) | Hepatotoxicity at high doses | Sustained-release forms riskier |
| Nicotinamide | Issues at multi-gram doses | Some individuals more sensitive |
Key Uncertainties Remaining
Despite encouraging short-term data, several critical questions remain unanswered:
Long-term continuous use: Years of daily NMN supplementation has not been rigorously studied. Cumulative effects on liver tissue remain unknown.
Pre-existing liver conditions: Effects in people with fatty liver, viral hepatitis, cirrhosis, or heavy alcohol use are largely unexplored.
Drug interactions: Interactions with polypharmacy (multiple medications) have not been systematically tested. This is particularly relevant for medications processed through the liver.
Cancer considerations: People with active malignancy or high cancer risk should only consider NMN in consultation with an oncologist, given theoretical concerns about NAD+ supporting cell survival and DNA repair.
Right now, NMN should be considered experimentally promising but not fully characterized, especially for liver outcomes. The health effects over decades of use simply aren’t known.
Verification and Authentication: Ensuring NMN Supplement Quality
With the growing popularity of NMN supplements, ensuring product quality has become more important than ever. Not all supplements are created equal, and the effectiveness—and safety—of NMN depends heavily on the purity and accuracy of what’s inside the bottle. Verification and authentication are essential steps to confirm that NMN supplements contain the correct amount of nicotinamide mononucleotide and are free from harmful contaminants.
The gold standard for evaluating NMN supplements is the double-blind, placebo-controlled clinical trial. These rigorous studies are designed to confirm both the efficacy and safety of NMN, providing reliable data on potential benefits such as increased insulin sensitivity, improved aerobic capacity, and support for age related diseases. Clinical trials not only help establish the science behind NMN but also set benchmarks for what consumers should expect from high-quality products.
Unfortunately, the quality of NMN supplements on the market can vary widely. That’s why verification successful authentication—through third-party lab testing and transparent reporting—is crucial. Reputable brands will provide certificates of analysis, confirming that their products have been independently tested for purity, potency, and safety. This level of verification gives consumers confidence that they are choosing a supplement that is both effective and safe.
By prioritizing NMN supplements that have undergone thorough verification and are backed by clinical trials, consumers can better support their health and well-being. As NMN research advances, ongoing authentication and scientific validation will remain essential to confirm the efficacy and safety of these promising supplements. Always look for products that are supported by science, tested for quality, and transparent about their manufacturing practices—because when it comes to your health, quality is essential.
Practical Considerations: Using NMN Responsibly With Liver Health in Mind
If you’re considering NMN supplements, your personal liver health status, current medications, and existing conditions should factor heavily into your decision. The current state of NMN research doesn’t yet support widespread recommendations for therapeutic use.
Who Should Be Especially Cautious
People with known liver conditions should speak with a hepatologist or primary care clinician before using NMN or any high-dose NAD+ precursor. This includes those with:
Non-alcoholic fatty liver disease (NAFLD)
Hepatitis B or C
Cirrhosis of any cause
Autoimmune hepatitis
Elevated liver enzymes from other causes
History of drug-induced liver injury
The same caution applies to pregnant or breastfeeding individuals, adolescents, and people on complex medication regimens—especially hepatotoxic drugs such as certain antifungals, anti-tuberculosis medicines, or chemotherapy.
A Cautious Approach for Healthy Adults
For otherwise healthy adults who wish to experiment with NMN after careful consideration:
Start low: Begin at the lower end of doses tested in clinical trials (e.g., 250–300 mg/day administered orally)
Avoid self-escalation: Don’t increase to very high doses without medical supervision
Limit initial duration: Consider limiting trial use to 8–12 weeks, mirroring available study durations
Consider professional oversight: Work with a healthcare provider who can monitor your response
Monitoring Strategies
For those using NMN under medical oversight, the following monitoring approach makes sense:
Baseline testing before starting:
Liver panel (ALT, AST, ALP, GGT, bilirubin)
Renal function tests
Fasting lipids and glucose
Follow-up testing:
Repeat the panel after 8–12 weeks
Look for any notable changes from baseline
Stop and seek medical advice if:
Liver enzymes rise significantly above baseline
Symptoms like jaundice (yellowing of skin/eyes) develop
Dark urine appears
Unexplained fatigue occurs
Right upper abdominal discomfort develops
Choosing Products Wisely
Where NMN supplements remain legally available, product quality varies significantly. Impurities—not NMN itself—could pose liver risks. Look for:
Transparent third-party testing certificates
Testing for identity and purity
Testing for contaminants (heavy metals, residual solvents)
Good manufacturing practice (GMP) certification
Clear labeling of dose and form
The efficacy and safety of any supplement depends partly on what’s actually in the bottle matching what’s on the label.

Lifestyle Strategies to Support NAD⁺ and Liver Health Without Supplements
Before reaching for any supplement, consider that there are non-supplement ways to support both NAD+ biology and liver health. These approaches are generally lower risk and beneficial regardless of whether you ever take NMN.
Regular physical activity Exercise training upregulates NAD+ biosynthesis enzymes and improves liver fat content and insulin sensitivity. The gold standard recommendation of at least 150 minutes per week of moderate aerobic capacity exercise provides measurable benefits. Studies in amateur runners and other active populations show improved metabolic markers compared to sedentary controls.
Weight management and diet A diet rich in whole foods—especially vegetables, fiber, and unsaturated fats—reduces fatty liver risk. Some foods naturally contain small amounts of NMN or related precursors (edamame, broccoli, cucumbers, avocados, cow’s milk), though these contribute minimally to supplement-level dosing. The dietary approach supports overall liver health regardless of NAD+ considerations.
Limiting alcohol intake Chronic drinking directly depletes hepatic NAD+ and injures liver cells. Reducing alcohol consumption protects liver function and supports the body’s natural NAD+ maintenance systems.
Sleep and circadian rhythm NAD+ metabolism is tightly linked to the body’s clock and liver metabolic cycles. Adequate sleep and consistent sleep-wake schedules contribute to healthy NAD+ regulation without any supplementation.
These lifestyle factors affect far more than just NAD+ levels—they address the root causes of many age related diseases and metabolic conditions. Disease prevention through lifestyle modifications remains more evidence-based than supplement interventions for most people.
Key Takeaways and Future Research Directions
What the Current Data Shows
Human studies administering up to 1,200–1,250 mg/day of NMN for 4–12 weeks have not shown clear liver toxicity in healthy adults
Clinical parameters including ALT, AST, and other liver enzymes remained within normal ranges in published trials
Animal studies often point toward improved liver metabolism and resilience, but at much higher relative doses than humans typically take
NMN appears to support SIRT3 activation and Nrf2-mediated antioxidant defenses in aged mice, providing potential mechanisms for liver protection
Long-term safety, high-dose use, and effects in people with liver disease or patients on multiple medications remain largely unknown
Research Priorities Going Forward
The NMN research field needs:
Larger, longer trials: Multi-year studies including participants with metabolic syndrome or early NAFLD, with detailed liver imaging and histology where ethically appropriate
Comparative studies: Head-to-head research comparing NMN versus other NAD+ precursors focusing specifically on hepatic and renal safety endpoints
Metabolomics work: Studies tracking downstream NAD+ metabolites (MNA, 2-PY, 4-PY) and their relation to liver and cardiovascular outcomes in humans
Special populations: Research in people with existing liver conditions to understand potential risks and benefits in these groups
Interaction studies: Systematic evaluation of how NMN interacts with common medications metabolized by the liver
The Bottom Line
As of 2024, NMN should be viewed as an experimental longevity-related compound with encouraging—but incomplete—liver safety data. The compound has shown remarkable results in some animal studies and appears well tolerated in short-term human trials. However, “no clear toxicity signal in limited studies” is not the same as “proven safe for long-term use.”
For anyone considering NMN, collaboration with healthcare professionals is strongly encouraged—especially for individuals with liver concerns, those taking multiple medications, or people with metabolic conditions like Alzheimer’s disease or diabetes that may affect how the body processes supplements.
Perhaps most importantly, foundational lifestyle changes remain central to both liver and overall metabolic health. The potential benefits of any supplement should be weighed against the proven benefits of exercise, healthy diet, limited alcohol, and adequate sleep. These interventions support NAD+ biology, protect your liver, and address the underlying factors that contribute to age-related metabolic decline.
The science of NMN continues to evolve. What we know today may expand significantly in the coming years as more data from ongoing research becomes available. Until then, informed caution remains the wisest approach.
Further Reading
Explore more articles related to this topic:
- Is NMN Safe for Your Liver? Analyzing the Latest 2026 Hepatotoxicity Data
- NMN and Heart Health: Can It Reduce Arterial Stiffness and Blood Pressure?
- Benefits Of NMN Supplements: Science-Backed Pros, Limits, And Safety
- Can I Get Enough NMN From Food?
- Can NMN Improve Muscle Strength in Adults Over 50?
- NMN and Alcohol: Does Drinking Deplete NAD+ and Negate Your Supplement?
- NMN Drug Interactions: Does NMN Interact with Statins, Blood Pressure Meds, and Antidepressants?