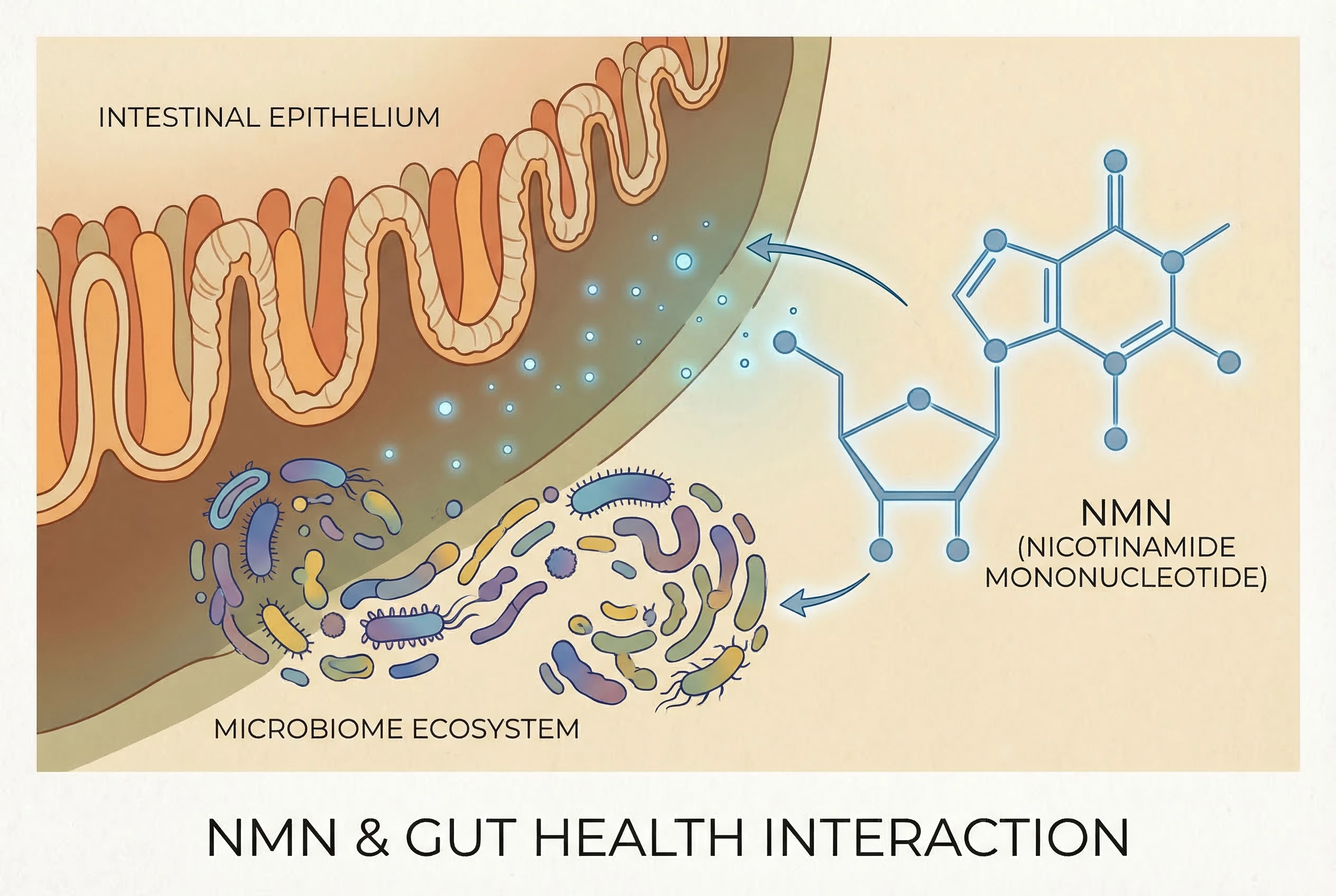
The intersection of nmn and gut health with cellular metabolism has become one of the more intriguing areas of modern biology. This article is for health-conscious individuals, researchers, and anyone interested in the science behind NMN and gut health. Understanding this relationship may help inform future approaches to digestive wellness and longevity. As interest in longevity-focused supplements grows, questions about NMN and gut health are surfacing more frequently among researchers and health-conscious individuals alike.
NMN, NAD+ and the Gut – Key Takeaways Upfront
If you’re reading this, you’re likely asking a straightforward question: “What does NMN do to my gut and microbiome, and is it safe?” This section provides direct answers before diving into the details.
At a glance:
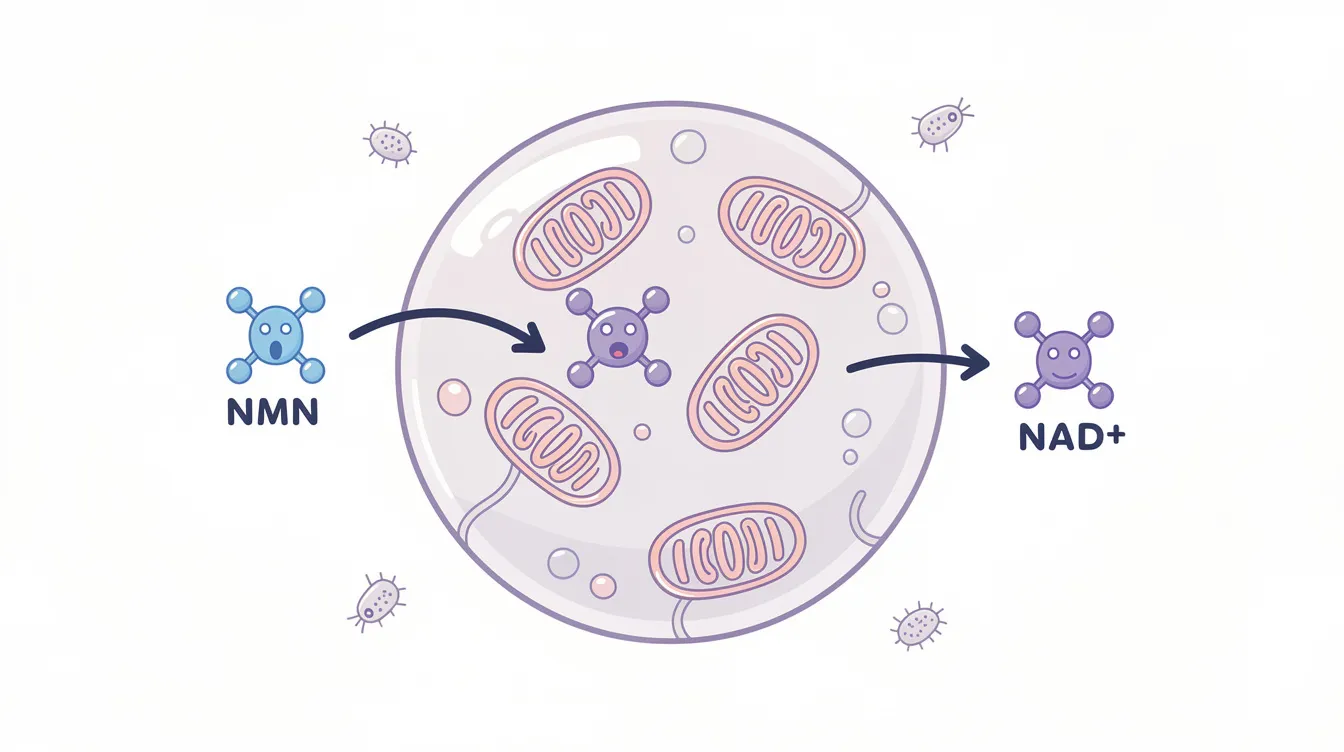
NMN (nicotinamide mononucleotide) is a precursor to NAD+ (nicotinamide adenine dinucleotide), a molecule involved in energy production, dna repair, and cellular stress responses throughout the human body.
NMN is emerging as a potent modulator of gut health due to its ability to increase NAD+ levels, supporting cellular repair and energy metabolism in the digestive tract.
Animal studies and early human data suggest NMN interacts with gut microbiota and intestinal barrier function, but the evidence is still emerging and primarily preclinical.
NMN has a prebiotic-like effect, modulating the gut’s microbial ecosystem.
Gut bacteria can transform NMN into other vitamin B3-related compounds—including nicotinic acid—that may influence how much NAD+ your body actually produces from supplementation.
Most detailed mechanistic findings come from mice and ex vivo (laboratory) studies. Human trials focused specifically on gut health endpoints remain limited as of 2024.
NMN is not approved to treat, prevent, or cure any gut disease. Supplementation should not replace medical care for digestive conditions.
Safety profiles from short-term human trials suggest tolerability at common doses, though long-term gut-specific safety data are sparse.
Before starting any supplement regimen—especially if you have inflammatory bowel disease, irritable bowel syndrome, or other chronic digestive conditions—discuss your plans with a healthcare professional. The research is promising but incomplete.
Summary of Main Findings:
NMN increases beneficial bacteria such as Akkermansia, Bacteroides, Lactobacillus, Bifidobacterium, and Firmicutes.
NMN decreases harmful bacteria including Staphylococcus, Bilophila, Oscillibacter, and Desulfovibrionaceae.
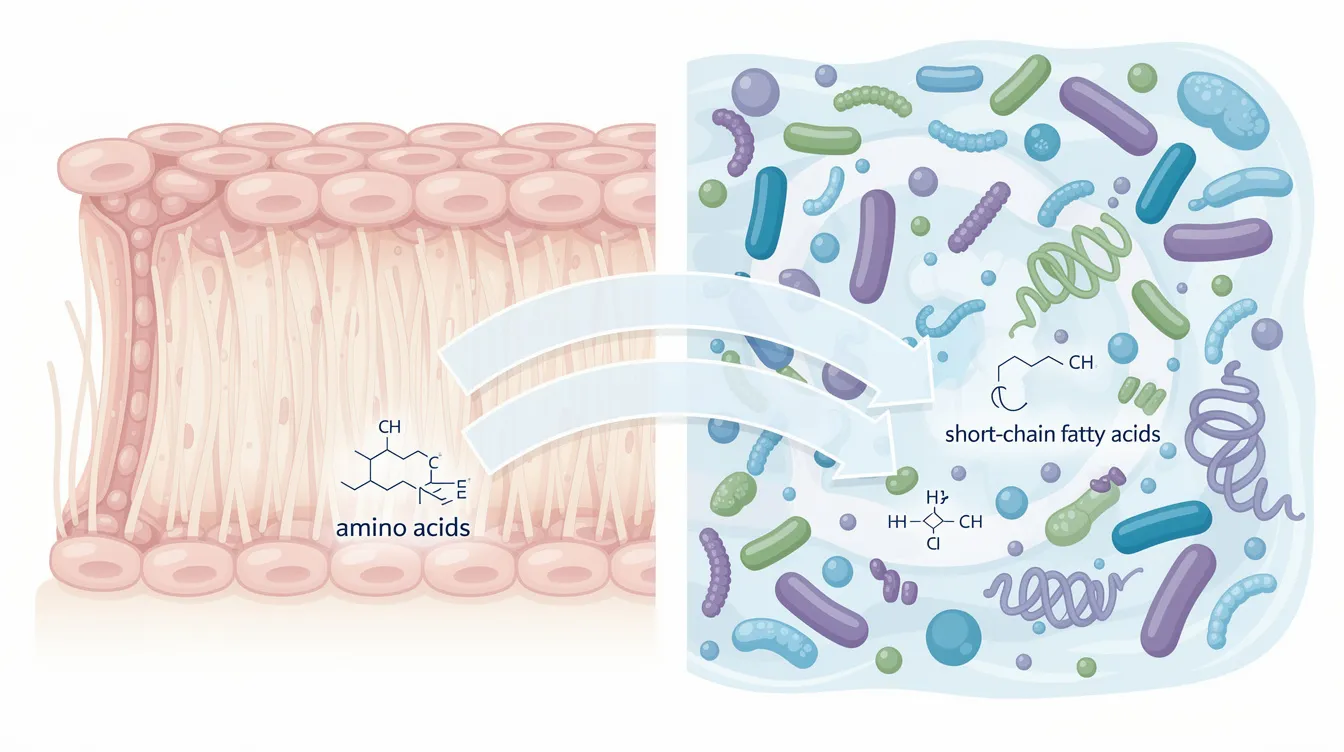
NMN boosts the production of short-chain fatty acids (SCFAs) like butyrate and propionate, which support gut function and reduce inflammation.
NMN improves gut barrier function and upregulates tight junction proteins.
NMN reduces inflammatory cytokines and markers in the gut.
NMN restores colon length and improves intestinal morphology in IBD models.
NMN helps repair gut flora dysbiosis and supports the integrity of the intestinal lining.
What Are NMN and NAD+? A Quick Refresher
Nicotinamide mononucleotide is a vitamin B3-derived compound found naturally in small amounts in foods like edamame, broccoli, and avocados, as well as within mammalian cells themselves. While dietary sources typically provide only 1-2 mg per 100 grams, supplements offer concentrated doses ranging from 250 mg to 1000 mg daily.
NMN serves as a building block that the body uses to synthesize nicotinamide adenine dinucleotide, commonly known as NAD+. This coenzyme is essential for:
Energy production in mitochondria, where it participates in converting nutrients to cellular fuel
DNA repair processes that maintain genomic integrity
Sirtuin activation and other NAD+-dependent enzyme functions linked to aging and metabolism
General cellular stress responses that help cells survive and adapt to challenges
NMN is emerging as a potent modulator of gut health due to its ability to increase NAD+ levels, supporting cellular repair and energy metabolism in the digestive tract.
NAD+ levels naturally decline with age and during periods of chronic stress. This decline has motivated substantial research into NAD+ “boosters” like NMN and its cousin, nicotinamide riboside (NR).
How does the body make NAD+?
There are three primary synthesis routes:
| Pathway | Starting Point | Key Enzymes | Contribution |
|---|---|---|---|
| Salvage pathway | Nicotinamide (NAM) | NAMPT, NMNAT | ~85% of NAD+ |
| De novo pathway | Tryptophan | Multiple enzymes | Variable |
| Preiss Handler pathway | Nicotinic acid (NA) | NAPRT | Context-dependent |
| In addition to these, the de novo pathway and microbiome-mediated enzymatic reactions, such as deamidation, are integral to NAD+ biosynthesis. The role of d f in these pathways highlights how gut bacteria and their enzymes (like PncC and PncA) can influence NAD+ metabolite levels. |
Here’s what makes this relevant to gut health: the gut and its resident bacteria can influence which pathway predominates. When you take NMN orally, it doesn’t simply get absorbed intact—gut microbiota play an active role in processing it.
Why the Gut Microbiome Matters in NAD+ Metabolism
The gastrointestinal tract hosts trillions of microorganisms—bacteria, archaea, fungi, and viruses—collectively known as the gut microbiome. These residents don’t just passively occupy space. They digest dietary components, produce metabolites, train the immune system, and interact with virtually every aspect of host biology.
Certain gut bacteria can:
Use vitamin B3-related compounds (nicotinamide, nicotinic acid, NMN, NR) as nutrients for their own metabolism
Convert one form into another, such as transforming nicotinamide to nicotinic acid via bacterial enzymes called nicotinamidases
Compete with host tissues for NAD+ precursors, potentially affecting how much reaches your cells
Evidence from related research:
A 2022 study published in Cell Death & Disease examined how intestinal microbiota influence NAD+ metabolism in leukemia mouse models. Researchers analyzed cell death, protein expression, and metabolite levels using laboratory techniques to interpret the effects of gut microbiota on NAD+ metabolism. The study also compared the effects of different NAD+ precursors and treatment outcomes to highlight differences in NAD+ metabolism and bacterial activity. Researchers found that gut bacteria converted dietary nicotinamide to nicotinic acid, which altered how NAD+-lowering drugs performed. While this study focused on cancer treatment rather than supplements, it highlighted a broader principle: microbes actively reshape vitamin B3 metabolism in ways that can affect therapeutic outcomes.
This doesn’t mean NMN is harmful. Rather, it illustrates that gut microbiota are active participants—not passive bystanders—in how NAD+ precursors are processed.
The presence of different bacterial species, their relative abundance, and their metabolic activity all contribute to this complex picture. Your gut isn’t just a tube for absorption; it’s a processing plant that can modify what you consume before your tissues ever see it.
How NMN Interacts With the Gut Microbiome: What the Science Shows
Deamidation and Microbial Processing
Most detailed evidence about NMN-microbiome interactions comes from mouse studies and in vitro or ex vivo experiments. Human data focused specifically on gut outcomes remain relatively scarce as of 2024. That said, the mechanistic work is illuminating.
Key findings from 2022-2023 research:
Researchers have examined how bacteria “deamidate” NMN—essentially stripping off an amino group to convert it into nicotinic acid mononucleotide (NaMN). This shifts the balance between “amidated” compounds (like NMN itself) and “deamidated” compounds (like NaMN and nicotinic acid).
A 2023 study published in Science Advances tracked what happens after mice were treated with NMN via oral gavage. In some experiments, mice were also treated with antibiotics to deplete their microbiome and study the effects on NAD+ metabolism:
Early response (15-30 minutes): NAD+ levels peaked in the small intestine, likely through direct uptake via the salvage pathway
Later response (around 3 hours): Liver NAD+ increased via the Preiss Handler pathway, dependent on microbial conversion of NMN to nicotinic acid and related deamidated forms
When researchers depleted gut bacteria using an antibiotic cocktail, the results shifted dramatically:
| Condition | Deamidated Metabolites | Amidated Forms (NR, NMN) |
|---|---|---|
| Normal microbiome | Elevated | Lower |
| Antibiotic treatment | Reduced 50-70% | Elevated |
| These findings suggest that oral NMN is largely processed by intestinal bacteria into nicotinic acid before being used for liver NAD+ synthesis. The microbiome essentially acts as a metabolic gatekeeper. Certain bacteria may also protect cells by maintaining NAD+ levels, which could influence the efficacy of NMN and related compounds. |
What does this mean practically?
The data suggest a complex host-microbe partnership. Your gut bacteria shape how much NAD+ your body ultimately produces from NMN and related compounds. This may explain some of the variability people report in their responses to nmn supplementation.
Understanding these pathways could inform future medicine and therapeutic strategies targeting NAD+ metabolism and gut health.

Short-Term NMN Exposure: Ex Vivo Data
Some research groups have incubated human stool samples with NMN for approximately 24 hours to observe which bacteria thrive or decline. These ex vivo experiments provide clues about potential microbiome effects.
Key observations from stool culture studies:
Overall bacterial diversity sometimes decreased over the 24-hour exposure period
Bacteroides species—a common bacterial genus in the human gut—showed increased relative abundance in the presence of NMN
Levels of short chain fatty acids (SCFAs), particularly propionate, increased under these conditions
Important methodological caveat:
Human digestion typically moves material through the small intestine in roughly 6-8 hours. A 24-hour continuous exposure in a laboratory dish doesn’t perfectly mimic real digestion. The concentration and duration of NMN exposure exceed what would occur physiologically.
Ex vivo findings are useful preliminary signals, but they cannot directly predict clinical outcomes in people taking NMN supplements in normal doses.
What are SCFAs and why do they matter?
Short chain fatty acids—primarily acetate, propionate, and butyrate—are produced when gut bacteria ferment dietary fiber and other substrates. These metabolites:
Fuel colonocytes (colon cells)
Support gut barrier integrity via tight junction proteins
Act as signaling molecules influencing inflammation and metabolism
The observation that NMN exposure can increase propionate production in vitro is intriguing but needs confirmation in living systems.
Chronic NMN Supplementation in Mice
Longer-term studies in aged mice provide additional perspective. One chronic supplementation study administered approximately 400 mg/kg/day of NMN for 8-10 weeks and assessed microbiome composition using 16S rRNA sequencing.
High-level findings:
No major changes in overall microbial diversity indices (alpha and beta diversity remained stable)
Some shifts in individual species abundance at the genus level, but community structure remained largely intact
Firmicutes populations stayed stable, and no dramatic dysbiosis occurred
What this might mean for you:
Unlike broad-spectrum antibiotics—which can devastate microbial communities—NMN in this mouse model appeared to act more like a metabolic modulator than a community-disrupting agent. The microbiome composition wasn’t dramatically altered.
Limitations to keep in mind:
Mouse physiology and microbiota differ from humans in important ways
Mouse-equivalent dosing (400 mg/kg) far exceeds typical human supplement doses
The study assessed composition, not necessarily function (metabolite output, gene expression)
Results suggest NMN supplementation doesn’t behave like an antibiotic in terms of microbiome disruption, but more research is needed to understand functional implications.
NMN, Gut Barrier Function and Inflammation: Emerging Data
NMN and Intestinal Barrier Integrity
The intestinal barrier is a single layer of epithelial cells sealed by tight junction proteins—including claudin-1, occludin, and E-cadherin. This barrier performs a critical balancing act: allowing nutrient absorption while preventing gut bacteria, toxins, and undigested food particles from entering the bloodstream.
When this barrier becomes compromised—sometimes called “leaky gut” in popular parlance—it can contribute to local and systemic inflammation. Maintaining barrier integrity is a key aspect of gut health.
NMN in IBD Models: Effects on Inflammation and Barrier Function
The DSS-induced colitis mouse model:
A 2022 study published in Actions examined NMN’s effects in mice with chemically induced colitis (using dextran sodium sulfate, or DSS). This model mimics some features of inflammatory bowel disease.
Reported outcomes:
| Measure | Colitis Mice (Untreated) | Colitis Mice + NMN Treatment |
|---|---|---|
| Body weight loss | Significant | Attenuated |
| Colon length | Shortened | Better preserved |
| Histological damage | Severe | Improved |
| Tight junction proteins | Reduced | Upregulated |
| Permeability markers | Elevated | Decreased |
| Inflammatory cytokines (TNF-α, IL-6) | High levels | Reduced |
| Additionally, nmn treatment increased the relative abundance of beneficial bacteria associated with gut health, including Akkermansia, Lactobacillus, Verrucomicrobia, and Firmicutes. |
Critical context:
These results come from a specific mouse model of inflammatory bowel disease—not from humans with ulcerative colitis or Crohn’s disease. Animal models often exaggerate effects and don’t always predict human outcomes.
NMN may influence inflammation and barrier function in animal intestines, but it should not be viewed as a stand-alone treatment for IBD. Any therapy decisions must involve a gastroenterologist.
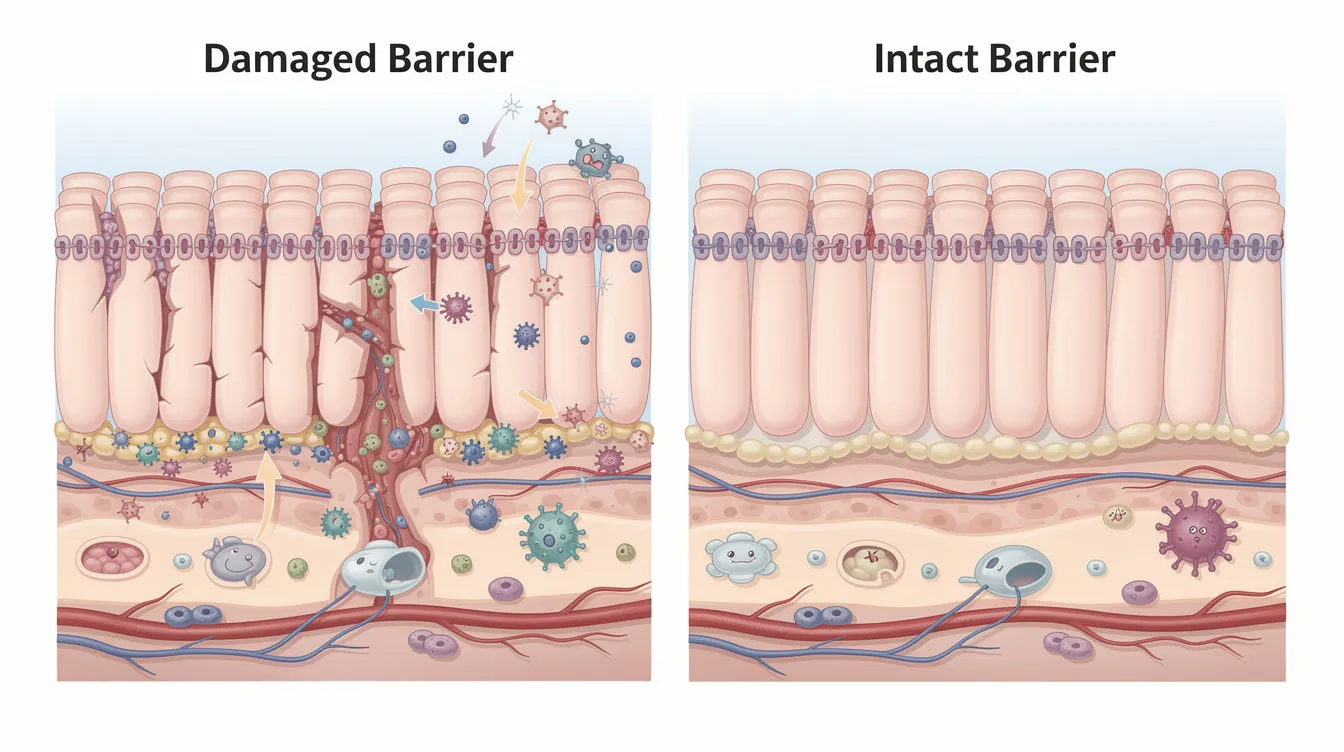
Linking NAD+ to Barrier Integrity Mechanistically
How might NAD+ actually support gut barrier health? Several mechanisms have been proposed, though the relative importance of each remains under investigation.
Conceptual connections:
Energy for repair: Maintaining tight junctions is energy-intensive. NAD+ fuels mitochondrial ATP production, which intestinal epithelial cells need for constant renewal and repair.
Enzyme regulation: NAD+ is required for enzymes like sirtuins (SIRT1, SIRT3) and PARPs that regulate inflammation, cell death pathways, and stress resilience.
Stress adaptation: Adequate NAD+ may help cells maintain tight junction proteins when exposed to inflammatory insults or oxidative stress.
In the DSS colitis work and related studies, raising NAD+ levels via precursors coincided with better barrier markers and reduced inflammatory cytokines. However, it remains uncertain whether the main driver is:
Direct metabolic support of epithelial cells
Indirect effects mediated through shifts in intestinal microbiota
Some combination of both and other mechanisms
The biology is complex, and researchers continue to identify the precise causal pathways.
Cell Death Regulation: A New Angle on NMN and Gut Health
Recent research has begun to shed light on a fascinating aspect of NMN treatment: its ability to regulate cell death in the gut, with potential implications for overall gut health. The lining of the gastrointestinal tract is constantly renewing itself, and a delicate balance between cell survival and cell death is essential for maintaining a healthy barrier against pathogens and supporting nutrient absorption.
Nicotinamide adenine dinucleotide (NAD+)—the molecule at the heart of NMN supplementation—plays a pivotal role in this process. By supporting energy production and DNA repair in gut epithelial cells, NAD+ helps these cells withstand stress and recover from damage. NMN supplementation has been shown to boost NAD+ levels in the human body, which may, in turn, enhance the resilience of the gut lining.
One intriguing mechanism involves the de novo pathway, an alternative route for NAD+ synthesis. NMN treatment has been found to increase the production of nicotinic acid mononucleotide, a key intermediate in this pathway. This shift may help support cellular energy needs and promote the survival of beneficial bacteria within the gut microbiota. In fact, studies suggest that NMN supplementation can increase the relative abundance of beneficial bacteria, further supporting gut health.
The interplay between NMN, cell death regulation, and the gut microbiota highlights the complexity of the gut ecosystem. While NMN’s ability to promote DNA repair and modulate cell death is promising, the precise mechanisms—especially how these effects translate to improved gut health in humans—are still being unraveled. As research continues, understanding how NMN influences both the cells lining the gut and the bacteria that inhabit it may open new avenues for therapeutic strategies aimed at maintaining a healthy gastrointestinal tract.
Antibiotic Treatment Impact: NMN and the Microbiome Under Stress
Antibiotic treatment is well known for its ability to disrupt the gut microbiome, often leading to a dramatic shift in the relative abundance of both beneficial and harmful bacteria. This disruption can have far-reaching effects on gut health, and it also raises important questions about how NMN supplementation works when the microbiome is under stress.
Studies have shown that NMN supplementation can increase the abundance of beneficial bacteria, such as Bacteroides, which are associated with positive gut health outcomes. However, when antibiotics are introduced—especially broad-spectrum antibiotic cocktails containing agents like streptomycin, gentamicin, and ciprofloxacin—the composition of the gut microbiota can change significantly. These changes may reduce the efficacy of NMN supplementation, as the bacteria responsible for processing NMN and supporting its conversion to key metabolites may be depleted.
The impact of antibiotic treatment on NMN’s benefits is an area of active investigation. Some research suggests that after antibiotic treatment, the gut’s ability to convert NMN into nicotinic acid and other important intermediates is diminished, potentially limiting the positive effects of NMN on NAD+ levels and gut health. This highlights the importance of the gut microbiota in mediating the effects of NMN supplementation and suggests that the timing and context of supplementation—such as during or after antibiotic use—may influence outcomes.
As with many aspects of gut biology, more research is needed to fully understand how antibiotic treatment interacts with NMN supplementation. For now, these findings underscore the importance of maintaining a healthy and diverse gut microbiome, especially when considering supplements aimed at supporting cellular metabolism and gut health. If you are undergoing antibiotic treatment and considering NMN supplementation, it’s wise to consult with a healthcare professional to ensure your approach is both safe and effective.
Potential Benefits: How NMN Could Influence Gut Health (Hypotheses, Not Promises)
Important disclaimer: The following points summarize hypotheses and early findings, not established benefits. No NMN product is approved by regulatory agencies to treat, prevent, or cure any gut disease. This section reflects areas of scientific interest, not medical recommendations.
Areas under investigation:
| Potential Effect | Evidence Status | Key Caveats |
|---|---|---|
| Microbial composition shifts | Mouse and ex vivo studies show some taxa changes | Human microbiome effects unconfirmed |
| SCFA production changes | Ex vivo data show propionate increases | Duration and concentration exceed physiological exposure |
| Barrier protein expression | Mouse colitis models show upregulation | Disease model may not translate to healthy humans |
| Local inflammation reduction | Preclinical work shows cytokine decreases | No human IBD trials with gut endpoints |
| What’s currently missing: |
Randomized controlled human trials with primary endpoints on gut barrier function
Human microbiome sequencing before and after NMN supplementation
Clinically relevant gut outcomes (calprotectin for inflammation, zonulin for permeability)
Long-term safety data in diverse populations
These represent signals worth exploring rather than actionable clinical guidance. The possibility that NMN influences gut health is being actively investigated, but verification successful completion of human trials remains pending.
Risks, Unknowns and Practical Considerations
Key Unknowns
While NMN has been generally well tolerated in short-term human studies aimed at systemic NAD+ levels, long-term data and detailed gut-focused safety data are limited. Here’s what potential users should consider.
Long-term effects of chronic NAD+ elevation on gut microbial ecosystems in diverse human populations
Impact on people with existing gut conditions (IBD, IBS, celiac disease, SIBO, history of colon surgery)
Effects of combining NMN with other NAD+ boosters (NR, nicotinamide, nicotinic acid) on the microbiome
Whether baseline microbiome composition predicts response to supplementation
Practical Considerations
| Factor | What to Know |
|---|---|
| Regulatory status | Varies by country—dietary ingredient in some regions, investigational new drug status in others (check local guidance) |
| Quality concerns | Purity issues exist; some products contain less than stated NMN. Look for third-party testing and certificates of analysis |
| Dosing uncertainty | Preclinical studies often use 300-500 mg/kg in mice, far exceeding human supplement doses of 250-1000 mg/day |
| GI tolerability | Short-term studies report mild GI upset in fewer than 5% of participants |
Interaction Considerations
Antibiotics can dramatically change microbiome composition and may alter NMN metabolism
The security of your supplement routine depends on understanding potential drug interactions
People with complex medication regimens or immune conditions should discuss any supplement use with their clinician
Questions to ask your healthcare provider before starting NMN:
Do I have any gastrointestinal diagnoses that might affect how I respond?
Will NMN interact with my current medications?
Am I pregnant, breastfeeding, or planning to become pregnant?
Am I enrolled in any clinical trials that might be affected?
What symptoms should prompt me to stop and seek evaluation?
Additionally, be wary of any website or security service making extravagant claims. Marketing language like “restores gut health” or “cures leaky gut” goes beyond current evidence. A security verification of any product’s claims against published research is prudent.
How to Support Gut Health While Research on NMN Continues
Established, evidence-supported strategies for gut health should take priority over experimental supplement approaches. While waiting for more definitive NMN research, these interventions have stronger backing:
Science-backed habits for gut health:
Diverse, fiber-rich diet: Whole grains, fruits, vegetables, and legumes nourish beneficial bacteria and increase SCFA production. Aim for approximately 30 grams of fiber daily.
Fermented foods: Yogurt with live cultures, kefir, sauerkraut, and kimchi can introduce helpful microorganisms if tolerated.
Limit ultra-processed foods: These often contain emulsifiers and additives that may disrupt the gut barrier in some individuals.
Moderate alcohol intake: Excessive alcohol can increase intestinal permeability and shift microbiome composition unfavorably.
Avoid unnecessary antibiotics: While sometimes essential, antibiotics can dramatically reduce microbial diversity. Use them only when prescribed.
Regular physical activity: Exercise has been associated with favorable microbial profiles in multiple observational studies.
Adequate sleep and stress management: Chronic stress can alter gut permeability and microbiota composition through the gut-brain axis.
Connecting lifestyle to NAD+ biology:
Many of these factors support mitochondrial function and metabolic resilience—the same systems influenced by NAD+-dependent pathways. Diet in particular shapes vitamin B3 intake through niacin and tryptophan, potentially affecting NAD+ production via the de novo pathway and Preiss Handler pathway.
Food sources of NAD+ precursors:
| Food | NMN Content | Other B3 Forms |
|---|---|---|
| Edamame | ~1-2 mg/100g | Niacin |
| Broccoli | ~1 mg/100g | Niacin |
| Avocado | Trace | Niacin |
| Mushrooms | Trace | Niacin |
| Dietary sources provide far less NMN than supplements, but a nutrient-rich diet supports NAD+ metabolism through multiple routes. Drinking water and overall hydration also support healthy digestion, though not directly related to NAD+. |
NMN is just one of several potential levers influencing cellular metabolism networks. You can meaningfully impact gut and metabolic health through everyday choices while scientists continue clarifying where NMN fits.
Looking Ahead: Key Research Questions on NMN and the Microbiome
The field is evolving rapidly. Several high-priority research questions remain unanswered, representing opportunities for future investigation.
Questions driving current research:
In humans, does regular NMN supplementation meaningfully change gut microbiome composition or function beyond normal day-to-day variation?
How do different doses and formulations of NMN (capsule, powder, co-formulation with nutrients) affect NAD+ levels and microbiota-derived metabolites in the gastrointestinal tract?
Are there specific microbiome “signatures” that predict who responds most strongly to NMN in terms of NAD+ boosting? For example, do individuals with higher baseline Akkermansia populations respond differently?
Could NMN or other NAD+ precursors be combined with dietary patterns or probiotics in future trials targeting gut barrier function or inflammatory markers?
What are the long-term safety implications of chronic NMN use for people with and without pre-existing gut conditions?
How do age, sex, diet, medications, and baseline microbiome composition modulate NMN’s effects?
Can findings from germ free mice studies help identify which specific bacteria mediate NMN’s effects?
Research approaches on the horizon:
Studies by groups including those led by researchers like Wang F, Chen Y, Shi X, and Wu X continue exploring NAD+ metabolism, microbiome interactions, and therapeutic strategies. While the authors declare ongoing interest in this field, independent verification of findings through replication studies remains essential.
Where to find updates:
Readers especially interested in this area can search public clinical trial databases for registered studies on NMN and NAD+ precursors. New trials are being launched and updated regularly, with some predicting gut-focused endpoints by 2027.
Closing perspective:
NMN represents a fascinating tool for probing the relationship between cellular metabolism and the gut ecosystem. The emerging picture—where gut microbiota actively process oral supplements, influence NAD+ production pathways, and potentially mediate some of NMN’s effects—adds nuance to simplistic narratives about “just taking a pill.”
For now, NMN should be viewed as a subject of active research rather than a proven gut health intervention. The development of better human data will help determine whether the promising signals from mouse models, ex vivo experiments, and early human trials translate into meaningful clinical applications.
In the meantime, focus on the basics: fiber, movement, sleep, and stress management. These remain the foundation of gut health—no matter what future research reveals about NAD+ precursors.
Further Reading
Explore more articles related to this topic:
- NMN and Alcohol: Does Drinking Deplete NAD+ and Negate Your Supplement?
- NMN for Diabetes: Can It Improve Insulin Sensitivity in Type 2?
- Health Benefits of NMN: What Science Really Shows
- NMN and the Liver: Is It Safe or Toxic? The Latest Data
- Best Mitochondrial Health Supplements for 2026: A Science-Backed Guide