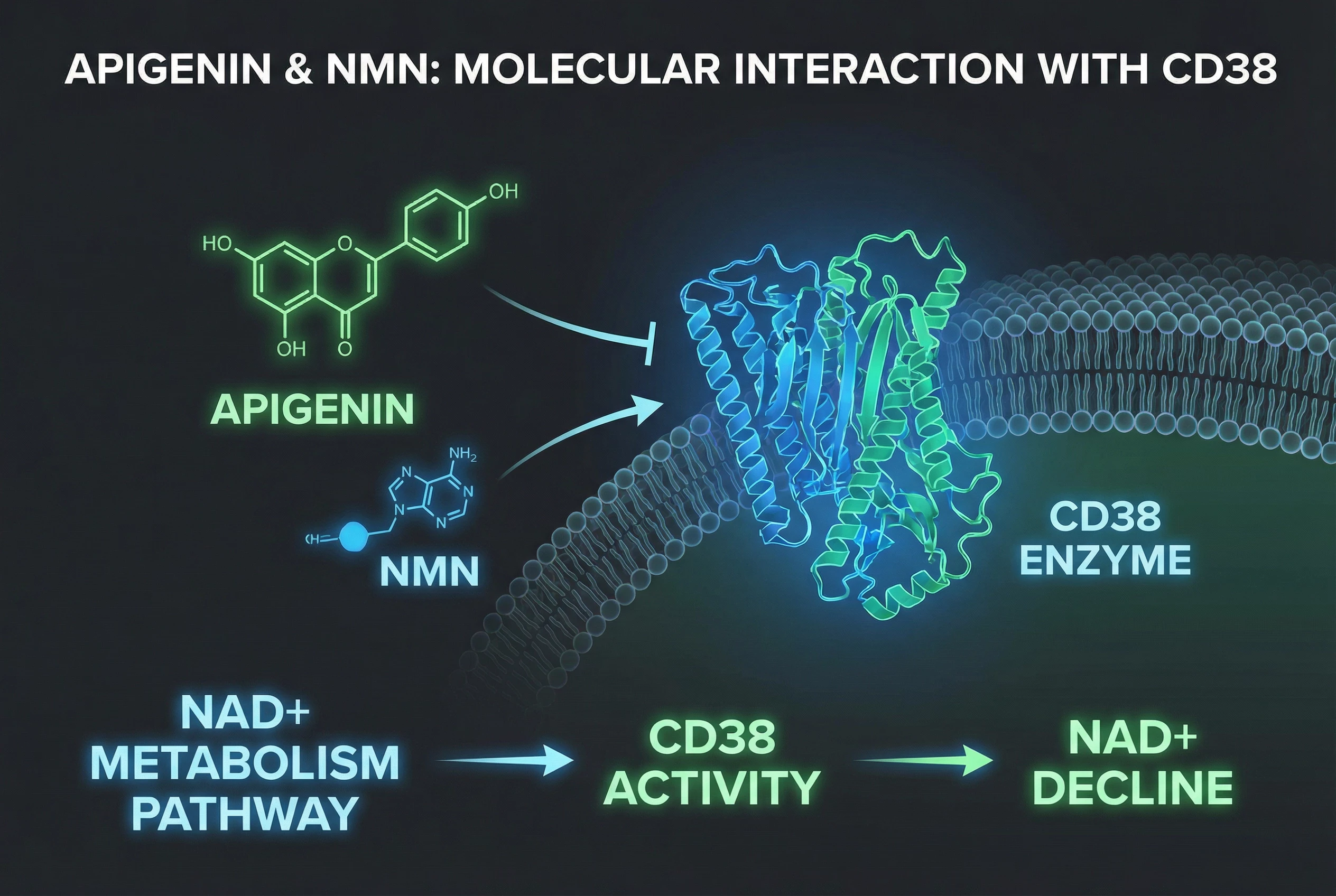
If you’ve spent any time exploring longevity research or NAD+ supplementation, you’ve likely encountered discussions about stacking different compounds for synergistic effects. One combination that’s generating significant interest is NMN paired with apigenin. But what’s the actual science behind this stack, and does it hold up to scrutiny?
This guide breaks down everything you need to know about the NMN and apigenin combination—from the fundamental biology of NAD+ metabolism to the preclinical evidence, practical considerations, and the substantial gaps that still exist in our understanding. Whether you’re a researcher, a curious self-experimenter, or simply someone trying to make sense of the supplement landscape, this deep dive will help you separate promising hypotheses from proven facts.
Quick Overview: Why Combine NMN and Apigenin?
Nicotinamide mononucleotide, commonly known as NMN, is a naturally occurring molecule that serves as a direct precursor to nicotinamide adenine dinucleotide (NAD+). Think of it as a building block your cells use to manufacture one of the most important coenzymes in human biology. NAD+ participates in over 500 enzymatic reactions, from energy production to DNA repair, and its levels appear to decline progressively as we age.
Apigenin is a different kind of molecule altogether. It’s a flavonoid—a type of plant compound—found abundantly in chamomile, parsley, celery, and other common foods. While apigenin has been studied for various biological effects, including anti-inflammatory activity and sedative properties, one area of particular interest is its apparent ability to modulate an enzyme called CD38 in laboratory settings.
The logic behind stacking these two compounds comes down to supply and demand. NMN works on the supply side of the equation: it feeds into the cellular machinery that produces NAD+. Apigenin, based on preclinical research, may work on the demand side by potentially slowing down one of the major pathways through which NAD+ gets consumed—specifically, the CD38 enzyme. In theory, if you’re both increasing production and reducing breakdown, you might achieve higher steady-state NAD+ levels than either approach alone.
Here’s a simple way to visualize the concept:
NAD+ Production Pathway: Tryptophan → Niacin → NMN → NAD+
NAD+ Breakdown Pathway: NAD+ → CD38 → cyclic adp ribose (cADPR) + ADPR
The stacking hypothesis positions NMN as an accelerator on the first pathway and apigenin as a brake on the second.
Important Safety and Policy Note
Before diving deeper, it’s essential to establish clear expectations. The evidence for this combination in humans is extremely limited. Most of what we know comes from animal studies and cell culture experiments. This article is purely informational and does not constitute medical advice.
If you’re considering any supplement regimen, especially one involving compounds that may affect fundamental cellular processes like NAD+ metabolism, you should consult with a qualified healthcare professional. This is particularly important if you have existing health conditions, take medications, or are pregnant or breastfeeding.
The supplement industry operates with minimal regulatory oversight, and product quality varies substantially. There is very little research on long-term effects of these compounds at supplemental doses in humans.
Understanding NAD+, CD38, NMN, and Apigenin
To understand why people stack NMN with apigenin, we need to look at how NAD+ is made and broken down, and where CD38 fits into this picture. These four concepts—NAD+, CD38, NMN, and apigenin—are interconnected in ways that form the biological basis for this supplementation strategy.
NAD+: The Central Player
Nicotinamide adenine dinucleotide (NAD+) is a coenzyme present in every living cell. It functions primarily as a redox cofactor, meaning it shuttles electrons during metabolic reactions. This makes it indispensable for processes like glycolysis, the TCA cycle, and oxidative phosphorylation—essentially, how your cells convert food into usable energy.
But NAD+ does more than just facilitate energy production. It serves as a substrate for several important enzyme families:
Sirtuins: NAD+-dependent deacetylases (like SIRT1 and SIRT3) that regulate protein acetylation, gene expression, and metabolic pathways
PARPs: Poly-ADP-ribose polymerases critical for DNA repair
CD38: An ectoenzyme that hydrolyzes NAD+ to produce signaling molecules
Age-Related NAD+ Decline
Research published in landmark reviews between 2016 and 2022 has documented that NAD+ levels decline progressively with age across multiple tissues, including muscle, liver, and brain. Some analyses of human tissue samples suggest NAD+ may drop by approximately 50% between young adulthood and old age. This age-related NAD decline has become a central focus of geroscience research.
CD38: The Major NAD+ Consumer
CD38 is a type II transmembrane glycoprotein expressed on immune cells, adipocytes, and various other tissues. What makes it particularly relevant to NAD+ biology is its function as an ecto-NADase—it sits on the cell membrane and efficiently hydrolyzes NAD+ into cyclic adp ribose (cADPR) and ADP-ribose (ADPR).
CD38’s Role in NAD+ Breakdown
These breakdown products aren’t useless; they play roles in calcium signaling and cellular communication. However, CD38’s high catalytic efficiency means it can rapidly deplete NAD+ pools, especially in tissues where CD38 expression is elevated.
Evidence Linking CD38 to Aging and Metabolic Dysfunction
Several lines of evidence connect CD38 to aging and metabolic dysfunction:
| Finding | Model | Key Observation |
|---|---|---|
| CD38 upregulation with age | Mouse tissues | 2-5 fold increase in aged vs. young |
| CD38 in obesity/inflammation | Human biopsies | Correlated with 30-50% lower NAD+ |
| CD38 knockout preservation | Mouse models | 2-3 fold higher NAD+ maintained |
| Extended lifespan | Some CD38 KO strains | 10-15% increase in median survival |
| These findings from previous studies suggest CD38 may be a significant driver of the age-related decline in NAD+ that researchers observe across species. |
NMN: The NAD+ Precursor
NMN occupies a critical position in what’s called the salvage pathway—the recycling route through which cells regenerate NAD+ from nicotinamide. The enzyme NAMPT converts nicotinamide to NMN, and then NMNAT enzymes convert NMN to NAD+.
NMN in Research
David Sinclair’s research group at Harvard published influential work around 2013-2016 demonstrating that NMN administration in mice could restore NAD+ levels in aged tissues, improve vascular endothelial function, and enhance exercise capacity. These animal models provided the initial excitement around NMN supplementation.
Human studies have followed, though they remain relatively small and short-term. Trials from 2020-2022 using oral NMN at doses of 250-500 mg/day have reported improved insulin sensitivity in prediabetic cohorts and favorable safety signals. One 2022 randomized controlled trial with 36 participants showed a 38% increase in blood NAD+ and 12% improvement in aerobic capacity over 12 weeks.
However, NMN doesn’t appear to directly inhibit CD38. In fact, CD38 can hydrolyze NMN itself, which raises questions about efficiency in tissues with high CD38 expression.
Apigenin: The Natural Molecule Under Investigation
Apigenin (4’,5,7-trihydroxyflavone) is a natural molecule classified as a flavone—a subclass of flavonoids. It’s found in:
Chamomile: 3-5 mg per gram in dried flowers
Parsley: ~45 mg per 100g fresh
Celery: ~20 mg per 100g
Biological Activities of Apigenin
Beyond its presence in foods and chamomile tea, apigenin has attracted research attention for several biological activities, including neuroprotection in animal models of Alzheimer’s disease, anti-inflammatory effects, and potential anti-cancer properties in cell culture studies.
For the purposes of this discussion, the most relevant finding is that apigenin appears to act as a potent inhibitor of CD38 in laboratory settings. A 2013 study published in Diabetes demonstrated that apigenin treatment reduced CD38 activity in dose-dependent fashion, with an IC50 (concentration needed for 50% inhibition) of approximately 10-50 μM against human CD38.

The Science of CD38 Inhibition: Why It Matters for NAD+ and Aging
CD38 has emerged as a central NAD+ hydrolase that becomes more active in aged and metabolically stressed tissues. Understanding why inhibition of CD38 has attracted research interest requires looking at the downstream consequences of elevated CD38 activity.
CD38 Expression Increases with Age and Inflammation
Multiple lines of evidence show that CD38 expression rises in aged tissues and during chronic inflammation:
Aged mouse kidneys (24 months vs. 3 months) show approximately 3-fold higher CD38 expression
Adipose tissue and inflammatory cells in obese mice display elevated CD38 alongside reduced NAD+
Human biopsies from individuals with metabolic diseases show similar patterns
This upregulation correlates with markers of metabolic dysfunction, including altered glucose and lipid homeostasis and increased markers of reactive oxygen species production.
Functional Consequences of Elevated CD38
When CD38 activity increases, several downstream effects follow:
NAD+ depletion: Direct conversion of cellular NAD into breakdown products
Altered calcium signaling: cADPR and ADPR are calcium-mobilizing messengers
Mitochondrial dysfunction: NAD+/NADH ratio imbalances (dropping from ~10:1 to ~3:1 in high-glucose states) impair oxidative phosphorylation
Sirtuin impairment: Lower NAD+ means less substrate for SIRT3, leading to hyperacetylation of mitochondrial enzymes like IDH2 and SOD2
Increased oxidative stress: 2-3 fold elevation in reactive oxygen species observed in models
These changes connect CD38 to multiple age-related diseases and tissue damage patterns observed in aging and metabolic syndrome.
What Happens When CD38 Is Reduced?
Preclinical models using either genetic deletion or pharmacological inhibition of CD38 have produced notable results:
| Intervention | Model | Outcome |
|---|---|---|
| CD38 knockout | Mice | Heart NAD+ doubled; 20% improved ejection fraction post-ischemia |
| CD38 knockout | Diet-induced obesity model | Resistance to obesity development |
| Pharmacologic inhibition (apigenin, 78c) | Obese mice | 50-100% boost in NAD+; 30% increased mitochondrial respiration |
| Live-cell imaging work published in 2023-2024 using NR analogues has confirmed CD38’s surface localization and dynamic hydrolysis activity, providing visual confirmation of its role in NAD+ metabolism in living cells. |
Translation Challenges
Despite these encouraging findings from animal studies, translation to human supplementation strategies remains speculative. Key open questions include:
No direct CD38 activity assays have been performed in human supplementation trials
The degree of inhibition needed for clinical benefit is unknown
Whether partial inhibition (20-50% as might occur with dietary apigenin) is sufficient without disrupting CD38’s legitimate physiological roles
CD38 isn’t solely a “bad actor”—it plays important roles in immune function and cellular calcium signaling. Complete or excessive inhibition could have unintended consequences.
Apigenin as a CD38 Modulator
Apigenin is a flavonoid with multiple biological targets, one of which appears to be CD38 based on preclinical research. The flavonoid apigenin has been studied across various contexts, from cancer cells to neurodegenerative disease models, but the CD38 connection is particularly relevant for those interested in NAD+ support.
Key Evidence: Diabetic Kidney Disease Models
Some of the most detailed work on apigenin and CD38 comes from studies in diabetic kidney disease models. Research using Zucker Diabetic Fatty rats (a model of metabolic syndrome and type 2 diabetes) examined oral apigenin administration at approximately 20 mg/kg, given 5 days per week for 4 weeks.
Effects Observed in Diabetic Animals
The results demonstrate several notable effects in diabetic animals:
Renal CD38 mRNA and protein reduced by 40-60%
NAD+/NADH ratio improved from 2.5 to 5.8
SIRT3 target acetylation (ace-IDH2) decreased by approximately 50%
Fibrosis markers (collagen IV) reduced by 35%
Oxidative DNA damage marker (8-OHdG) decreased by 45%
Pro-inflammatory gene expression (TNF-α) reduced by 50%
Importantly, these effects were observed specifically in diabetic rats—lean controls showed minimal changes, suggesting context-dependent efficacy. Apigenin treatment appeared most beneficial in metabolically stressed conditions.
Cell Culture Findings
Complementary work in HK-2 cells (human proximal tubular cells from the American Type Culture Collection) grown under high glucose conditions (25 mM) showed similar patterns:
High glucose increased CD38 and lowered NAD+/NADH
10 μM apigenin for 48 hours normalized CD38 (reduced by ~60%)
NAD+/NADH ratio doubled
Mitochondrial reactive oxygen species decreased by 40%
SIRT3 activity restored, reducing mitochondrial enzyme acetylation
These results demonstrate apigenin’s effects in a human cell model of tubular cell damage, lending some relevance beyond rodent physiology. CD38 siRNA (genetic knockdown) produced similar outcomes, suggesting the CD38 pathway is genuinely involved.
Broader Apigenin Research
Beyond kidney models, apigenin has been studied in various contexts:
Neurodegeneration: In APP/PS1 Alzheimer’s disease models, 40-50 mg/kg oral apigenin reduced microglial CD38 and amyloid-β plaques in mice, suggesting potential protective effects in neurodegenerative disease contexts.
Sleep and Anxiety: Human studies using chamomile extract (containing apigenin among other compounds) have shown improvements in sleep biomarkers and generalized anxiety disorder symptoms. Postpartum women drinking chamomile tea showed 15-20 minute improvements in sleep latency. However, these studies used multi-component extracts, making it difficult to isolate apigenin’s specific contribution.
Metabolic Health: Animal models suggest apigenin increases insulin sensitivity and improves glucose and lipid homeostasis, potentially through CD38-independent mechanisms as well.
Critical Limitations
Several important caveats apply to this research:
Dosing discrepancy: Rodent doses of 10-50 mg/kg translate to approximately 100-200 mg in humans via allometric scaling—substantially higher than typical 50 mg supplements
No isolated human trials: Clinical studies have used chamomile extract, not isolated apigenin for CD38 endpoints
Context dependency: Benefits appear most pronounced in metabolically stressed states, raising questions about relevance for healthy individuals
Limited mechanistic clarity: Apigenin affects multiple pathways; attributing effects solely to CD38 inhibition oversimplifies the biology
Bioavailability, Metabolism, and Dosing Considerations for Apigenin
Absorption and Metabolism
Most dietary apigenin occurs as glycosides (sugar-attached forms) rather than free aglycone. When consumed:
Only an estimated 5-25% is absorbed in the small intestine (based on rat ileum studies)
Rapid phase II metabolism (glucuronidation, sulfation) converts absorbed apigenin into conjugates
These conjugates constitute up to 80% of plasma flavonoids
Unabsorbed apigenin reaches the colon, where gut microbiota convert it to phenolic metabolites like phloretic acid
Solubility Challenges
Apigenin aglycone is poorly water-soluble (<1 μg/mL), which limits absorption. Some supplement manufacturers use liposomal formulations, which have shown 3-fold improved Cmax (peak blood levels) in rodent studies.
Food Sources vs. Supplements
| Source | Approximate Apigenin Content |
|---|---|
| Dried parsley | ~45 mg per gram |
| Fresh celery | ~20 mg per 100g |
| Chamomile tea (cup) | 0.8-1.2 mg |
| Dried chamomile flowers | 3-5 mg per gram |
| Typical supplements | 20-100 mg per capsule |
Dosing Considerations
Due to limited human pharmacokinetic data, any specific dose recommendations would be speculative. Animal studies typically use 10-50 mg/kg, which far exceeds what most people obtain from diet or standard supplements.
Open Research Questions
Several areas need further studies:
Form optimization: Is aglycone or glycoside form better for absorption and tissue distribution?
Microbiome variability: Bifidobacterium species may enhance apigenin metabolism 2-fold; individual gut composition matters
Dosing pattern: Does chronic intake more durably affect CD38 expression compared to acute bolus dosing?
Tissue targeting: Which tissues achieve meaningful apigenin concentrations at practical oral doses?
NMN: Building the NAD+ Pool
NMN is a key intermediate in the NAD+ salvage pathway, produced from nicotinamide via the enzyme NAMPT. This salvage pathway accounts for approximately 70% of NAD+ synthesis flux in most tissues, making it the dominant route for maintaining cellular NAD+ levels.
Preclinical Evidence
The scientific interest in NMN supplementation began with animal models showing significant changes in NAD+ levels and downstream markers:
Intraperitoneal NMN at 300 mg/kg in mice tripled liver NAD+ within 15 minutes
Chronic oral administration maintained 2x elevations for extended periods
Muscle showed 40-60% NAD+ improvements with chronic dosing
Age-impaired glucose uptake improved from 50% to 95% of young levels in skeletal muscle
Vascular endothelial function and exercise capacity improved in aged mice
These findings from David Sinclair’s laboratory and others established NMN as a viable NAD+ precursor with biological activity in animal models.
Human Trial Data
Human studies have emerged since 2019, though they remain limited in size and duration:
| Study | Participants | Dose | Duration | Primary Outcomes |
|---|---|---|---|---|
| Japanese trial (2020) | 66 | 300 mg/day | 60 days | 25% improved muscle insulin signaling |
| RCT (2022) | 36 | 250 mg/day | 12 weeks | 38% blood NAD+ increase; 12% aerobic capacity |
| Prediabetes trial (2023) | Various | 600 mg/day | Variable | 20% muscle NAD+ increase (biopsy confirmed) |
| These results demonstrate bioavailability and suggest potential benefits for insulin sensitivity and NAD metabolism, though larger and longer trials are needed to confirm efficacy and establish optimal dosing. |
NMN Is Not a CD38 Inhibitor
An important clarification: NMN’s documented mechanism is as a precursor feeding into NAD+ synthesis, not as a direct inhibitor of CD38. In fact, the opposite may be true—CD38 can hydrolyze NMN with a KM of approximately 50 μM, meaning it efficiently processes NMN as a substrate.
In tissues with elevated levels of CD38 (such as inflamed or aged kidney), CD38 may catabolize 20-30% of supplemented NMN before it can be converted to NAD+. This substrate competition provides part of the rationale for considering CD38 inhibition alongside NAD+ precursor supplementation.
How NMN and CD38 Interact in Experimental Systems
Enzyme Kinetics
In vitro enzyme assays reveal important details:
CD38 hydrolyzes NAD+ efficiently (kcat/Km ~10^7 M^-1 s^-1)
NMN hydrolysis occurs at 10-20% the rate of NAD+ hydrolysis
Nicotinamide riboside (NR), another NAD+ precursor, is comparatively resistant to CD38
Recent structural work (PDB ID 8VAU, 2023) shows NR can form a covalent inhibitory complex with CD38 (Ki <1 μM)
Cellular Studies
In HEK293 cells overexpressing CD38, NMN supplementation raised NAD+ only 1.5-fold compared to 3-fold in wild-type cells with normal CD38 levels. This suggests that high CD38 activity can partially offset the benefits of NMN supplementation.
The Stacking Hypothesis
These kinetic observations form the basis for the combination strategy:
NMN provides raw material for NAD+ synthesis
CD38 can consume both NMN and the resulting NAD+
Reducing CD38 activity (with apigenin or other modulators) might preserve more NMN-derived NAD+
Models suggest 20-50% additional NAD+ preservation when CD38 is inhibited
However, this hypothesis remains untested in clinical trials. Communities experimenting with this combination are operating based on mechanistic logic rather than direct human evidence.
Cautionary Notes
Enzyme kinetics observed in test tubes and living cells don’t automatically translate to clear clinical benefits:
Tissue distribution of both compounds matters
Timing and dosing relationships are unknown
Individual variation in CD38 expression and activity could affect outcomes
The endoplasmic reticulum and other cellular compartments may have distinct NAD+ pools
The Rationale for Stacking NMN and Apigenin
The stacking strategy seeks a “two-lever” approach to NAD+ homeostasis: one lever pushes NAD+ up (NMN supplementation), while the other tries to slow NAD+ breakdown (apigenin via CD38 modulation).
A Systems Perspective
NAD+ homeostasis depends on multiple factors:
Synthesis inputs:
Dietary precursors (tryptophan, niacin, nicotinamide)
Salvage pathway activity (NAMPT, NMNAT enzymes)
De novo synthesis capacity
Consumption outputs:
CD38 hydrolysis
PARP activity (especially during DNA damage)
Sirtuin-mediated deacetylation
Other NAD+-consuming enzymes
Lifestyle modulators:
Sleep quality (poor sleep associated with reduced NAD+)
Exercise (boosts NAMPT activity)
Caloric intake and metabolic state
Circadian rhythm alignment
NMN and apigenin address only a subset of these levers. They don’t directly affect PARP activity, sirtuin expression, or the lifestyle factors that strongly influence NAD+ biology.
Theoretical Advantages
Based on preclinical data, the potential benefits of combining these approaches include:
Higher baseline NAD+ pools through simultaneous supply increase and consumption decrease
Improved mitochondrial redox balance via restored NAD+/NADH ratios
Enhanced sirtuin activity through greater substrate availability
Tissue-specific benefits particularly in organs with high CD38 (kidney, adipose, immune tissue)
The diabetic kidney studies are illustrative: apigenin normalized NAD+/NADH and restored SIRT3 activity in rat models. Independent evidence shows NMN raises NAD+ and supports sirtuin function in other contexts. Combining both approaches could theoretically provide complementary benefits.
The Evidence Gap
Explicitly stated: as of early 2026, there are no published randomized human trials testing NMN and apigenin together for:
NAD+ levels
CD38 activity
Sleep outcomes
Metabolic parameters
Aging biomarkers
The combination remains a hypothesis derived from mechanistic reasoning and extrapolation from separate lines of research. Self-experimenters are essentially conducting n=1 trials without rigorous controls.
Questions Worth Considering
For readers and researchers interested in this area, several thoughtful questions deserve attention:
Timing: Should NMN and apigenin be taken together, or would staggered dosing be more effective? CD38 expression may have circadian patterns.
Tissue specificity: Which tissues would benefit most? Kidney effects are well-documented in models, but brain and muscle data are less developed.
When is inhibition desirable?: CD38 plays roles in immune function and calcium signaling. Is partial inhibition always beneficial, or might it impair certain physiological processes?
Dose-response relationships: What degree of CD38 inhibition is achievable with practical apigenin doses? Is it enough to meaningfully affect NAD+ in humans?
Individual variation: CD38 expression varies substantially between individuals. Would baseline CD38 status affect who might benefit most?
Potential Goals of an NMN + Apigenin Stack (Hypothesis-Driven)
Based on online discourse in longevity communities and forums, common user goals for this combination include:
Healthy Aging Support
The primary motivation for many is addressing cellular aging through NAD+ optimization. The reasoning connects NAD+ to:
Sirtuin-mediated metabolic regulation
Mitochondrial function maintenance
DNA repair capacity
Inflammatory gene expression modulation
Whether raising NAD+ through supplementation actually slows aging in humans remains unproven, but the theoretical connections are plausible.
Metabolic Resilience
Given apigenin’s effects on glucose and lipid homeostasis in animal studies, and NMN’s impact on insulin sensitivity in small human trials, some users hope the combination might support healthy sleep and metabolic function, particularly for those with low grade inflammation or metabolic dysfunction risk factors.
Sleep and Recovery
The sleep angle draws on multiple lines of evidence:
NAD+ and circadian biology: NAD+ synthesis via NAMPT is under circadian control; NAD+ levels influence clock gene function through sirtuin activity
Sleep deprivation effects: Poor sleep has been associated with increased CD38 expression (up to 2-fold in some studies) and reduced NAD+
Apigenin’s sedative properties: Apigenin shows affinity for GABA-A receptors (IC50 ~10 μM), which may contribute to the traditional use of chamomile extract to improve sleep quality and support healthy sleep
Improved sleep is a commonly sought benefit of this stack, as poor sleep can accelerate aging and aging itself can reduce sleep quality. The role of the microbiome and lifestyle factors is also recognized in both sleep quality and aging processes.
However, the sleep benefits of chamomile likely involve multiple compounds and mechanisms beyond CD38 inhibition alone. Users hoping this stack will dramatically improve sleep should temper expectations.
Cognitive Performance
Some users extrapolate from animal studies showing apigenin’s effects in neurodegenerative disease models, hoping the combination might support brain health. NAD+ decline in neural tissue correlates with age, and restored NAD+ has shown benefits in some mouse models. Whether this translates to cognitive benefits in healthy humans is unknown.
It is important to note that high doses of either NMN or apigenin can cause mild gastrointestinal discomfort.
Realistic Expectations for Self-Experimenters
If you’re considering exploring this combination, think in terms of measurable, non-medical endpoints:
Subjective energy levels
Sleep duration and perceived quality
Simple fitness metrics (exercise recovery, endurance)
Heart rate variability (if tracking)
These are not validated biomarkers of NAD+ status or biological age. Blood NAD+ tests exist but correlate poorly (r~0.4) with tissue levels, limiting their utility for monitoring stack effectiveness.
Any self-experiment should be treated as exploratory, documented carefully, and undertaken with awareness of:
Placebo effects (substantial in subjective measures)
Day-to-day variability in how you feel
The limitations of consumer biomarker tests
Your inability to measure actual CD38 activity or tissue NAD+

Practical Considerations, Safety, and Open Questions
Anyone considering NMN or apigenin should first review their medical history and current medications with a healthcare professional. This is particularly important for conditions that might be affected by changes in NAD metabolism:
Diabetes or prediabetes (NAD+ and apigenin both affect glucose regulation)
Kidney disease (given the renal effects documented in models)
Cancer (NAD+ can fuel both healthy and cancer cells)
Autoimmune disorders (CD38 plays roles in immune function)
Pregnancy or breastfeeding (no safety data available)
NMN Safety Profile
Short-term human trials have generally reported favorable tolerability:
Mild GI symptoms in <5% of participants at doses up to 1g/day
Occasional flushing
No serious adverse events reported in trials up to 12 months
However, long-term safety data are sparse. NAD+ influences fundamental cellular processes, and the consequences of chronically elevated levels remain unknown.
Apigenin Safety Profile
Apigenin from dietary sources is generally recognized as safe. However, concentrated supplemental doses are less studied:
Potential concerns include:
Drug interactions: In vitro evidence suggests apigenin may inhibit CYP1A2 and potentially other drug-metabolizing enzymes, which could raise levels of caffeine, theophylline, and certain medications
Unknown chronic effects: Long-term high doses supplementation hasn’t been evaluated
Theoretical cancer considerations: While apigenin shows anti-cancer properties in cell studies, the NAD+ it might help preserve could theoretically benefit any rapidly dividing cells
Interaction Considerations
Both compounds could, in theory, influence:
Glucose metabolism (significant changes possible in sensitive individuals)
Vascular tone
Immune responses
In vulnerable populations, even subtle shifts might matter. Medical supervision is prudent for anyone with health conditions.
Framework for Stack Design
Without recommending specific doses (which would be inappropriate given the evidence gaps), here’s a general approach some self-experimenters consider:
Phase 1 (Weeks 1-4): Single Compound
Start with one compound (typically NMN) at conservative doses
Monitor for any adverse effects
Document baseline subjective metrics
Assess tolerability before adding complexity
Phase 2 (Weeks 5+): Add Second Compound
If Phase 1 is well-tolerated, introduce apigenin at low doses
Some prefer evening dosing for apigenin given its sedative effect potential
Continue monitoring and documenting
Phase 3: Adjustment
Based on personal response, adjust timing or doses
Consider periodic breaks to assess baseline vs. supplemented states
This is a hypothetical framework, not a clinical protocol. Individual responses vary, and professional guidance is advisable.
Lifestyle, Diet, and Microbiome Factors That Modulate NAD+ and CD38
Before focusing on supplements, it’s worth recognizing that lifestyle factors exert powerful effects on NAD+ biology and CD38 expression. Insights from molecular science research show that these factors influence NAD+ metabolism, enzymes like CD38, and related molecular mechanisms involved in aging and sleep regulation:
Exercise
A single bout of exercise can double NAMPT expression for 24 hours
Regular exercise training maintains elevated NAD+ synthesis capacity
May naturally suppress inflammatory CD38 upregulation
Sleep
Sleep deprivation increases CD38 expression and inflammatory cells activity
Disrupted circadian rhythms impair NAD+ synthesis timing
Quality sleep is probably more impactful than supplements for most people
Diet
Caloric balance affects NAD+ homeostasis
Polyphenol-rich diets (potentially providing 50 mg/day apigenin equivalents from food) may offer baseline CD38 modulation
Fiber supports microbiome diversity, which affects apigenin metabolism
Microbiome Considerations
Gut bacteria metabolize apigenin into secondary compounds
Akkermansia and Bifidobacterium species enhance flavonoid metabolism
Individual microbiome composition may substantially affect apigenin’s biological activity
Fermented foods and dietary fiber support beneficial bacterial populations
The core message: NMN and apigenin, if used, should be viewed as adjuncts to—not substitutes for—lifestyle factors that have robust evidence for supporting healthy aging and metabolic function.

Priority Hierarchy for NAD+ Optimization:
Core fundamentals: Quality sleep, regular exercise, stress management, circadian alignment
Dietary patterns: Whole foods, adequate protein, polyphenol-rich plant foods, appropriate caloric intake
Body composition: Maintaining healthy body weight and muscle mass
Microbiome support: Fiber, fermented foods, limiting unnecessary antibiotics
Experimental supplements: NAD+ precursors, CD38 modulators, and similar compounds
Skipping to level 5 while ignoring levels 1-4 is unlikely to produce meaningful results.
Future Directions and Research Gaps
The NMN and apigenin combination sits at an interesting intersection: mechanistically plausible, supported by preclinical data, but essentially untested in humans as a combined strategy. Several major questions need answers. To address these, future studies will require advanced research tools and standardized assays. Companies like Thermo Fisher and Thermo Fisher Scientific provide a range of kits and reagents that could facilitate such research, including those for mitochondrial extraction, protein analysis, and cell-based experiments.
Critical Unanswered Questions
Efficacy of combination: Does stacking NMN with apigenin meaningfully increase tissue NAD+ beyond NMN alone in humans? If so, by how much, and in which tissues?
Tissue specificity: Most preclinical work focuses on kidney, adipose, and immune cells. What about skeletal muscle, brain, cardiovascular tissue? Do relevant concentrations of apigenin reach these sites?
Safety of partial CD38 inhibition: CD38 serves legitimate physiological functions. What happens with chronic partial suppression over months or years? Are there immune consequences?
Optimal dosing: What apigenin dose is needed to achieve meaningful CD38 inhibition in humans? Current supplements may be far below effective levels based on allometric scaling from animal doses.
Types of Studies Needed
To advance this field, researchers should prioritize:
Randomized, placebo-controlled trials testing the combination vs. individual components vs. placebo
Tissue-level measurements going beyond blood NAD+ to measure NAD+ and NAD+/NADH ratios in biopsied tissue where feasible
CD38 activity assays in supplementation trials (currently not performed)
Apigenin pharmacokinetics in humans specifically measuring oral bioavailability, tissue distribution, and effects of chronic dosing
Mechanistic studies disentangling CD38-dependent from CD38-independent effects of apigenin
Long-term outcomes tracking not just biomarkers but clinical endpoints related to aging, sleep quality, metabolic health
Interdisciplinary Collaboration
Given apigenin’s broad potential footprint—renal injuries, neural protection, metabolic regulation, inflammatory modulation—collaboration between different research communities would be valuable:
Geroscience (aging mechanisms)
Nephrology (kidney function and diabetic kidney disease)
Neurology (neurodegenerative disease and cognitive aging)
Sleep medicine (sleep biomarkers and quality)
Immunology (CD38’s immune functions)
The authors declare that such collaboration could accelerate understanding of both therapeutic potential and risks.
Questions for Readers to Consider
As you evaluate this information, some reflective questions may be useful:
Is maximizing NAD+ always beneficial? Or might there be an optimal range, above which returns diminish or risks emerge?
How should you weigh promising preclinical data against absent long-term human outcomes? Animal models of multiple age-related diseases often don’t translate to human benefits.
What’s your personal risk tolerance? Self-experimenting with compounds that affect fundamental cellular processes isn’t without potential downsides.
Are your lifestyle fundamentals optimized? If sleep, exercise, and diet aren’t addressed, supplements are unlikely to compensate.
Can you track meaningful outcomes? Without good metrics, how will you know if the stack is doing anything?
A Balanced Perspective
The NMN and apigenin stack represents an intriguing, mechanistically grounded hypothesis. The logic is sound: NMN provides NAD+ building blocks; apigenin may reduce NAD+ breakdown via CD38 modulation. Preclinical data from animal studies support both individual components.
However, this remains an experimental approach. There are no combination trials in humans. Optimal dosing is unknown. Long-term safety of CD38 modulation is unstudied. Apigenin bioavailability at practical doses is questionable.
For those interested in exploring this space, proceeding cautiously makes sense:
Prioritize lifestyle factors with established benefits
Start with single compounds at conservative doses
Document your experience systematically
Consult healthcare professionals, especially if you have health conditions
Stay informed as new research emerges
Maintain realistic expectations about what supplements can achieve
The science of NAD metabolism and CD38 inhibition is advancing rapidly. What we know today will likely be refined substantially in coming years. The best approach is one that balances curiosity with appropriate caution, informed by evolving evidence and professional guidance.
Key Takeaways:
NMN is a NAD+ precursor that feeds into cellular energy metabolism; human trials show promising but preliminary results
Apigenin appears to inhibit CD38 in preclinical models, potentially reducing NAD+ breakdown
The stacking rationale addresses both supply (NMN) and demand (apigenin) sides of NAD+ homeostasis
Most evidence comes from animal models; human combination trials don’t exist
Lifestyle factors (sleep, exercise, diet) have stronger evidence for supporting healthy aging than any supplement
Self-experimentation should be approached cautiously, with medical guidance and realistic expectations
Further studies are needed to determine whether this combination offers meaningful benefits in humans
Whether you’re a researcher following the science or someone considering personal experimentation, the NMN and apigenin story illustrates both the promise and limitations of translating preclinical findings into practical health strategies. The hypothesis is worth watching as evidence develops—but it’s not yet ready for confident recommendations.
CD38 Expression in Alzheimer’s Disease
Emerging research has highlighted a significant role for CD38 expression in the progression of neurodegenerative diseases, particularly Alzheimer’s disease (AD). Studies have found that CD38 is overexpressed in the brains of individuals with AD, which has important implications for cellular health and cognitive function. Elevated CD38 activity leads to a marked decline in nicotinamide adenine dinucleotide (NAD+) levels—a molecule essential for maintaining mitochondrial function and supporting healthy sleep patterns.
Impact on Mitochondrial Function and Sleep
The reduction in NAD+ caused by increased CD38 expression contributes to mitochondrial dysfunction, which is a hallmark of Alzheimer’s pathology. This dysfunction impairs the cell’s ability to produce energy efficiently, leading to increased production of reactive oxygen species and heightened oxidative stress. These changes not only accelerate neuronal damage but also disrupt sleep quality, a common and troubling symptom in AD patients.
Animal Model Insights
Animal models have provided further insight into this relationship. In studies where CD38 was inhibited—either genetically or through compounds like apigenin—researchers observed improvements in mitochondrial function, reductions in oxidative stress, and even enhancements in sleep quality. Apigenin, in particular, has shown promise as a potent inhibitor of CD38, helping to preserve NAD+ levels and potentially offering neuroprotective effects.
While these findings are encouraging, further studies are needed to fully understand the therapeutic potential of targeting CD38 in Alzheimer’s disease. Ongoing research aims to clarify whether interventions that modulate CD38 expression and NAD+ metabolism can meaningfully impact disease progression, improve sleep quality, and support overall brain health in AD.
Nicotinamide Riboside and CD38
Nicotinamide riboside (NR) is another NAD+ precursor that has gained attention for its ability to boost NAD+ levels in living cells. Supplementation with NR has been shown to support glucose and lipid homeostasis, reduce the risk of metabolic syndrome, and enhance mitochondrial function—factors that are crucial for metabolic health and the prevention of multiple age-related diseases.
NR and CD38: A Dual Approach
The relationship between NR and CD38 is an area of active investigation. While NR supplementation can increase NAD+ availability, CD38 remains a major consumer of NAD+, and its elevated expression can limit the effectiveness of NAD+ precursors. Some studies suggest that combining NR with a CD38 inhibitor, such as apigenin, may amplify the benefits by both increasing NAD+ supply and reducing its breakdown. This dual approach could be particularly valuable in conditions like diabetic kidney disease, obesity, and neurodegenerative disorders, where both NAD+ depletion and CD38 overactivity are implicated.
Broader Benefits and Research Needs
Beyond metabolic health, NR has also been associated with reduced expression of inflammatory genes and improvements in sleep quality, potentially through its effects on NAD+ metabolism and CD38 modulation. These findings point to broader potential benefits for NR, especially when used in combination with CD38 inhibitors, though the precise mechanisms remain to be fully elucidated.
As with other NAD+ boosting strategies, further research is needed to determine the optimal use of NR in conjunction with CD38 inhibition. Understanding how these interventions interact in living cells and in clinical settings will be key to unlocking their full therapeutic potential for metabolic, inflammatory, and neurodegenerative diseases.
Further Reading
Explore more articles related to this topic:
- NMN and Resveratrol: Benefits, How They Work, What the Science Says
- The Longevity Stack: Why NMN and Resveratrol Are Better Together
- The Ultimate Longevity Trio: Stacking NMN, TMG, and Spermidine
- Why You Need TMG with Your NMN: The Truth About Methyl Depletion
- NMN and Urolithin A: The New Frontier of Mitochondrial Health in 2026