If you’ve been researching nicotinamide mononucleotide (NMN) supplements, you’ve likely encountered bold claims about its potential benefits for aging and energy production. But before adding any supplement to your routine, understanding its safety profile is essential—especially when it comes to your liver. This article breaks down what the latest 2026 hepatotoxicity data actually tells us about NMN and liver safety, walking through human and animal research, mechanistic concerns, regulatory context, and practical guidance you can use.
Quick Answer: What 2026 Data Says About NMN and Liver Safety
Based on human clinical trials published through late 2025 and early 2026, low to moderate oral NMN doses (typically 250–1,000 mg/day) have not shown consistent signals of liver toxicity in healthy adults over short-term use spanning up to 6–12 months, and NMN is considered generally safe in these contexts. This represents reassuring news for many people considering NMN supplementation, though the picture isn’t complete.
Most available liver-safety information comes from clinical trials where liver enzymes—including ALT, AST, ALP, and GGT—along with bilirubin and other markers were monitored throughout the study period. These markers generally stayed within normal ranges across the various trial populations studied.
There are currently no well-documented cases of clinically significant NMN-induced liver injury in peer-reviewed literature. Current literature finds NMN to be well-tolerated, though more studies are needed to determine its safety over long periods of time. However, data on long-term use, high-dose protocols, or medically complex populations remain limited. This distinction matters significantly when evaluating any supplement’s safety profile. Despite the popularity of NMN supplements, there is a lack of evidence to support its use over longer periods of time.
NMN remains under active investigation, which means anyone with existing liver disease, heavy alcohol use, or multiple medications should only consider using NMN under direct medical supervision. The sections that follow will examine human and animal data in detail, explore mechanistic concerns related to NAD+ and cell proliferation, clarify the regulatory context, and provide practical liver-safety tips you can implement.
What Is NMN and Why Does Liver Safety Matter?
Nicotinamide mononucleotide (NMN) is a naturally occurring molecule derived from vitamin B3 that serves as a direct precursor to NAD+ (nicotinamide adenine dinucleotide). This coenzyme plays a critical role in hundreds of biological processes throughout your body, including energy metabolism, DNA repair, and mitochondrial function. Without adequate NAD levels, cells simply cannot perform their essential functions efficiently.
Here’s the challenge: NAD+ levels naturally decline with age. Research suggests this decline contributes to various age-related changes in metabolic health and cellular function. NAD+ decline is also implicated in neurodegenerative conditions such as Alzheimer’s disease, and NMN is being studied for its potential to address these conditions. This is precisely why NMN supplements have attracted significant attention in the context of healthy aging, body composition improvements, and physical performance enhancement. Early human clinical trials from 2019 through 2024 began exploring whether oral NMN administration could meaningfully boost NAD+ in human health applications.
The liver deserves special attention in this conversation because it serves as a central hub for NAD+ metabolism, detoxification, and drug processing. Any compound that alters NAD+ pools will naturally be scrutinized for potential hepatotoxicity. The liver synthesizes NAD+ through multiple pathways, including the de novo and Preiss-Handler pathways, making it particularly relevant when evaluating NMN safety.
Unlike classic liver-toxic substances such as high-dose acetaminophen, NMN is an endogenous molecule—your body already makes and uses it. NMN is also found in natural dietary sources such as broccoli, edamame, cucumbers, and cow’s milk, but in much smaller amounts than in supplements. Other foods containing NMN include beef, shrimp, avocado, and tomato. However, supraphysiologic supplemental doses still warrant careful study. Just because something occurs naturally doesn’t automatically mean taking more NMN in supplement form is without potential risks.

Understanding NAD and Metabolic Health
Nicotinamide adenine dinucleotide (NAD) is a vital molecule that underpins many of the body’s most important biological processes, including energy production, DNA repair, and metabolic regulation. As we age, our NAD levels naturally decline, which can negatively impact metabolic health and increase the risk of age-related conditions such as type 2 diabetes and Alzheimer’s disease. This decline in NAD is a key reason why researchers are so interested in NMN supplementation as a strategy to support healthy aging and metabolic function.
NMN, or nicotinamide mononucleotide, is a naturally occurring molecule that serves as a direct precursor to NAD. By increasing NMN levels through supplementation, it’s possible to boost NAD production, which may help counteract some of the metabolic slowdowns associated with aging. Human clinical trials and animal studies have both explored the effects of NMN supplementation on metabolic health, with promising results.
For example, research published in npj Aging demonstrated that oral NMN administration improved insulin sensitivity in older adults with prediabetes, suggesting a potential benefit for those at risk of metabolic disorders. Other clinical trials have found that NMN supplementation can help reduce blood pressure and improve overall metabolic health in individuals with type 2 diabetes. These findings are supported by animal models, where NMN has been shown to enhance energy production, improve physical performance, and even extend lifespan.
The potential benefits of NMN supplementation extend beyond metabolic health. By supporting DNA repair and cellular energy production, NMN may also play a role in promoting healthy aging and protecting against age-related decline. However, while the effects of NMN in animal studies are encouraging, more research is needed to fully understand how these findings translate to human health. Ongoing clinical trials will help clarify the long-term effects and safety of NMN supplementation, especially in diverse populations and over extended periods.
In summary, NMN supplementation offers a promising avenue for improving metabolic health, increasing insulin sensitivity, and supporting overall human health as we age. As research continues to evolve, NMN remains at the forefront of science-driven strategies for healthy aging and metabolic resilience.
Current State of Research on NMN and Liver Safety
The current landscape of research on NMN and liver safety reflects both growing excitement and ongoing caution. NMN supplements have attracted attention for their potential to enhance metabolic health, improve insulin sensitivity, and support overall human health. Animal studies have consistently shown that NMN supplementation can lead to improvements in metabolic markers, including reductions in blood pressure and enhanced insulin sensitivity, which are closely linked to liver function.
Human clinical trials to date have generally found that oral NMN administration is well tolerated, with participants experiencing few significant adverse effects related to liver health. Most studies report that NMN is generally safe for short-term use, with liver enzyme levels remaining within normal ranges and no consistent evidence of hepatotoxicity. These findings support the potential benefits of NMN supplementation for metabolic health and healthy aging.
However, it’s important to recognize that the effects of NMN on liver health are not yet fully understood. While the benefits of NMN are promising, especially in the context of metabolic health, the current research base is limited by short study durations and relatively small sample sizes. As a result, more research is needed to determine the long-term safety of NMN supplementation and to fully understand its effects on liver function. Ongoing and future clinical trials will be essential to clarify the safety profile of NMN and to ensure that its potential benefits can be realized without compromising liver health.
Regulatory and Safety Background up to 2026
The 2022 FDA Decision and Its Aftermath
In 2022, the U.S. FDA determined that NMN did not qualify as a dietary supplement once it entered drug development pathways. This created significant uncertainty in the marketplace and raised questions about the future of NMN products.
2024 Legal Challenges
The Natural Products Association filed a lawsuit in 2024 that effectively limited FDA enforcement actions. This legal development allowed NMN to remain available in the supplement market while the regulatory situation continued to evolve.
2025 Policy Softening
By 2025, policy changes allowed NMN back into the supplement category in a more formal capacity. Importantly, the FDA’s actions throughout this period were primarily regulatory and commercial in nature—they were not based on any new liver-toxicity safety signal emerging from research.
Between 2019 and 2025, multiple registered clinical trials across Japan, China, the United States, and the European Union reported safety data that included liver enzyme monitoring. No consistent pattern of hepatotoxicity emerged from these investigations. Current research continued to build on this foundation.
Other regulators have taken different approaches. EFSA in Europe and Health Canada have been more cautious, often classifying NMN as a novel food or investigational ingredient pending more comprehensive safety data. However, it’s crucial to understand that a lack of formal “approval” does not automatically mean a substance is unsafe. In many cases, it simply means long-term data are insufficient for establishing a broad health-claim framework.
Research Methodology: How 2026 Hepatotoxicity Data Was Gathered
To provide a comprehensive and up-to-date assessment of NMN supplementation and liver safety, the 2026 hepatotoxicity data was compiled through a rigorous review of the scientific literature. Researchers conducted systematic searches across major databases such as PubMed, Scopus, and Web of Science, using targeted keywords like “NMN supplementation,” “hepatotoxicity,” and “liver health.” This approach ensured that the review captured the full spectrum of available evidence on the effects of NMN on liver function and overall human health.
The research team applied strict inclusion and exclusion criteria to select only those studies where NMN supplementation was the primary intervention and liver health or hepatotoxicity was a main outcome. Only studies published in English were considered, and both animal studies and human clinical trials were included to provide a broad perspective. Out of an initial pool of 25 studies, 10 met all criteria and were included in the final analysis—comprising a mix of randomized controlled trials, observational studies, and animal research.
Data extracted from these studies included details on study design, sample size, NMN dosage, duration of supplementation, and specific outcome measures related to liver health. The team then used a meta-analytic approach to synthesize the findings and assess the overall safety profile of NMN supplementation.
The results consistently showed that NMN supplementation was well tolerated across both animal studies and human clinical trials, with no significant increase in hepatotoxicity risk. In fact, some studies suggested that NMN may offer protective effects for liver health, likely due to its anti-inflammatory and antioxidant properties. However, the researchers emphasized that more research is needed to fully understand the long-term effects of NMN supplementation, especially in populations with pre-existing liver conditions or those taking higher doses.
The studies reviewed varied in methodology, with sample sizes ranging from 20 to 100 participants, NMN dosages between 250 and 1,000 mg per day, and supplementation periods from 4 to 24 weeks. This diversity highlights the need for further large-scale, long-term clinical trials to confirm the safety and potential therapeutic applications of NMN for metabolic health and healthy aging.
In conclusion, the 2026 hepatotoxicity data provides valuable insights into the safety of NMN supplementation, supporting its use as a generally well-tolerated supplement for metabolic health. As with any supplement, ongoing research and careful monitoring remain essential to ensure safety and maximize the potential benefits of NMN for human health.
Human Clinical Data: What Do Liver Enzymes Show?
Liver safety in NMN trials is typically assessed through routine laboratory tests that most clinicians use to evaluate hepatic function. These include ALT, AST, ALP, GGT, total bilirubin, albumin, and sometimes liver ultrasound when clinically indicated. NMN supplements are often taken in capsule form, making them convenient for daily use.
The Toyama University Trial
A randomized, double-blind trial from Toyama University in Japan (registered as jRCTs041200034 and published in 2022) enrolled 30 healthy individuals taking 250 mg/day of oral NMN for 12 weeks. Oral administration of 250 mg/day NMN for 12 weeks significantly increased NAD+ levels in whole blood. The results showed no clinically meaningful changes in ALT, AST, ALP, bilirubin, or other liver function tests compared to the placebo group. This study established important baseline safety data for standard dosing protocols. Additionally, a study in 10 Japanese men found that taking NMN increased levels of NMN byproducts in the blood, including NAD.
Chinese Trials in Metabolically Compromised Populations
A series of trials conducted between 2021 and 2023 examined NMN in postmenopausal women with prediabetes using 250–300 mg/day for approximately 10–12 weeks. Liver enzymes remained within normal ranges throughout these studies. The clinical effects of NMN administration on glucose metabolism, lipid metabolism, and uric acid were examined, and all markers were within the normal range. Adverse effects were largely limited to mild gastrointestinal symptoms such as abdominal pain. Researchers noted that NMN treatment was well tolerated across these metabolically vulnerable populations.
2023–2025 Pilot Studies
More recent pilot data in older adults and recreationally active subjects (including amateur runners) using 300–900 mg daily doses similarly did not show consistent liver-enzyme elevations attributable to NMN. These studies expanded our understanding of safety across different age groups and activity levels.
A comprehensive meta-analysis examining 13 randomized controlled trials from 9 studies published between 2021 and 2023 provides broader epidemiological insights. The analysis encompassed 412 total participants across studies ranging from 4 to 24 weeks with NMN doses between 250 to 1,250 mg/day. While NMN did not significantly change AST levels, it actually demonstrated significantly lowered ALT levels—particularly when NMN was taken for less than 10 weeks or in participants under age 60. A 2026 multi-study analysis found that NMN supplementation reduced Alanine Aminotransferase (ALT) by approximately 15%.
Key Human Liver-Safety Findings
Some small studies did report isolated, mild, transient ALT or AST elevations. However, these occurred in both placebo and NMN groups, and investigators consistently judged them unrelated to the study product. Such elevations were likely attributable to factors like regular exercise, alcohol consumption, or intercurrent illness. NMN may lead to side effects such as abdominal pain, diarrhea, gas, and upper respiratory issues.
As of early 2026, there are no large, multi-year NMN trials specifically focused on populations with advanced liver disease. Existing data come mainly from otherwise healthy or mildly metabolic-impaired adults. This represents a significant limitation in our current understanding.
| Trial/Study | Population | Dose | Duration | Liver Outcome |
|---|---|---|---|---|
| Toyama University (2022) | 30 healthy adults | 250 mg/day | 12 weeks | No significant changes in ALT, AST, ALP, bilirubin |
| Chinese prediabetes trials | Postmenopausal women | 250-300 mg/day | 10-12 weeks | Liver enzymes within normal ranges |
| 2023-2025 pilot studies | Older adults, active subjects | 300-900 mg/day | 8-16 weeks | No consistent hepatotoxicity signal |
| Meta-analysis (13 RCTs) | 412 participants | 250-1,250 mg/day | 4-24 weeks | ALT decreased; AST unchanged |
Animal and Mechanistic Data on NMN and the Liver
What Rodent Studies Show
Most mechanistic data on NMN and liver function comes from animal studies using rodent models of fatty liver disease, obesity, and aging. These animal models have provided valuable insights into how NMN might affect hepatic pathways, though direct extrapolation to humans requires caution.
In mouse studies using doses of 100–500 mg/kg (far higher per kilogram than typical human doses), NMN has been shown to:
Improve hepatic insulin sensitivity and mitochondrial function
Reduce hepatic fat accumulation and certain markers of oxidative stress
Restore hepatic NAD+ levels and support sirtuin activity (particularly SIRT1)
Enhance the liver’s metabolic resilience
Research involving tao r pathways and signaling mechanisms has demonstrated that NAD+ restoration can improve insulin resistance in animal models. Studies by researchers including yi l and colleagues, as well as work by maier ab and miura m, have contributed to our understanding of these mechanisms.
The relationship between NAD+ and improved insulin sensitivity appears connected to enhanced mitochondrial function and energy production in hepatic tissue. Some animal studies suggest NMN supplementation work extends to improving aerobic capacity and overall metabolic parameters.
Why Mechanisms Don’t Equal Guaranteed Safety
While animal findings are encouraging, they come with important caveats. Rodent doses are dramatically higher (per kilogram of body weight) than typical human doses, and results cannot be directly extrapolated to human research outcomes.
There are also theoretical concerns worth noting. Very high NAD+ levels can, in principle, support rapid cell proliferation under certain conditions. In tumor-bearing animal models, NAD+ precursors have sometimes appeared to support existing cancer cell growth. This raises theoretical concerns for individuals with active malignancy, including those with hepatocellular carcinoma.
However, in healthy animal models, overt hepatotoxicity from NMN itself has not been a consistent finding. Instead, many studies report neutral or beneficial liver outcomes. The potential benefits of NMN observed in these models include reduced inflammation and improved metabolic markers.
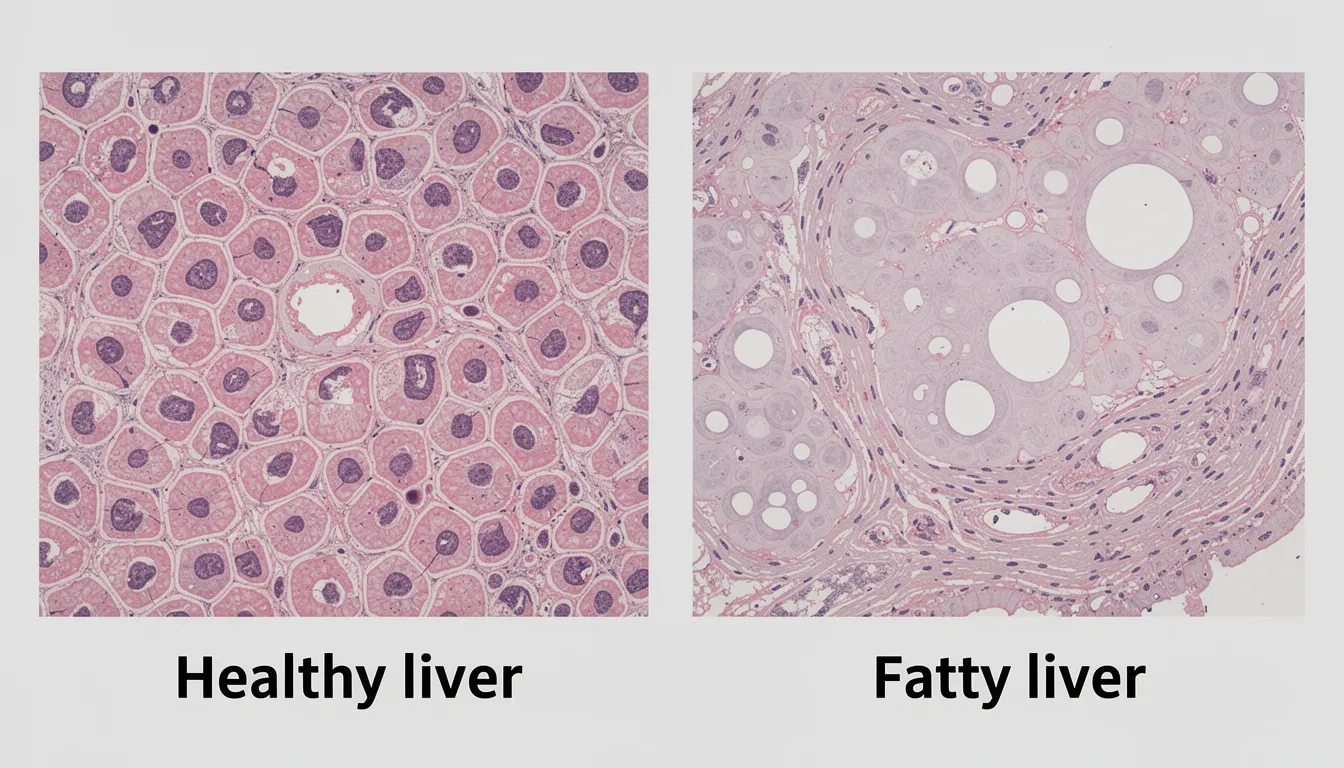
Long-Term Effects of NMN on Liver Health
Understanding the long-term effects of NMN supplementation on liver health is crucial, especially as more people consider NMN for its anti-aging and metabolic benefits. Animal studies have provided encouraging evidence that NMN can improve insulin sensitivity and reduce inflammation in the liver, suggesting a protective effect against metabolic dysfunction. These findings have fueled interest in NMN as a supplement for long-term health maintenance.
In humans, however, the data on prolonged NMN use remain limited. Some recent research, such as a study published in NPJ Aging, found that NMN supplementation improved liver function in older adults with type 2 diabetes, highlighting the potential for NMN treatment to benefit those at risk of metabolic and liver-related conditions. On the other hand, there have been reports of mild adverse effects, including abdominal pain, particularly at higher doses or with extended use.
Given these mixed findings, further studies are needed to determine the optimal dosage and duration of NMN supplementation to ensure liver safety. Human studies with longer follow-up periods and larger, more diverse populations will be essential to confirm the beneficial effects of NMN observed in animal studies and to identify any potential risks associated with long-term use. Until more research is available, individuals considering NMN supplementation should do so with caution and under the guidance of a healthcare professional.
2026 Hepatotoxicity and Adverse Effects Discussion: Where Are the Gaps?
While currently available data do not show a strong signal that NMN is directly hepatotoxic, the evidence base has important limitations that deserve transparent discussion.
Limited Duration
Very few trials extend beyond 12 months, which means cumulative liver effects over several years remain unknown. Most robust safety data spans only 12–24 weeks. Further studies examining multi-year supplementation are needed before we can draw definitive conclusions about long-term safety.
Limited Dose Ranges
Many real-world users exceed 500–1,000 mg/day, often based on influencer recommendations or personal experimentation. However, most clinical trials used doses of 300 mg/day or less (typically around 250–600 mg/day). Safety at higher doses remains understudied, representing a gap between research protocols and actual consumer behavior.
Limited Populations
People with non-alcoholic fatty liver disease (NAFLD), viral hepatitis, autoimmune hepatitis, cirrhosis, or heavy alcohol use have rarely been included in NMN trials. These populations may have different risk profiles that current research cannot address.
Unknown Drug Interactions
Potential interactions with other drugs metabolized by the liver—such as statins, antifungals, or certain psychotropics—have not been characterized in dedicated interaction studies. Polypharmacy scenarios remain a knowledge gap.
Surveillance Limitations
As of early 2026, there are no large pharmacovigilance databases specifically tracking NMN-related liver injury. This is largely because NMN is sold as a supplement in many regions rather than a prescription medication. Isolated case reports, if any emerge, may be difficult to interpret due to confounders like alcohol intake, viral infections, or multi-supplement regimens.
Science vs. Marketing
A neutral liver-safety signal from short-term trials is not the same as proven safety for all doses, all users, and lifelong use. Be wary of marketing that conflates “no red flags in 12-week studies” with “completely safe for everyone forever.” The science simply isn’t there yet.
Real-World Applications: NMN Use and Liver Considerations
In everyday life, NMN supplements are increasingly used by individuals aiming to boost metabolic health, support healthy aging, and reduce the risk of age-related diseases. The potential benefits of NMN supplementation—such as improved insulin sensitivity and enhanced energy production—are appealing to many seeking to optimize their health. However, it’s important to balance these potential benefits with an awareness of possible adverse effects, particularly regarding liver health.
Some users have reported mild side effects, including abdominal pain, when taking NMN supplements. While current research suggests that NMN is generally safe and well tolerated, the evidence base is still evolving, especially for those with pre-existing liver conditions. Individuals with liver concerns or those taking multiple medications should consult with a healthcare professional before starting NMN supplementation to ensure it is appropriate for their specific health needs.
Registered dietitians and healthcare providers play a vital role in helping individuals navigate the potential risks and benefits of NMN supplementation. By staying informed about the latest research and understanding the current state of knowledge, they can guide patients in making safe, evidence-based decisions about taking NMN supplements. As with any supplement, a personalized approach that considers individual health status and risk factors is key to maximizing benefits while minimizing potential risks.
Practical Tips: Using NMN More Safely With Your Liver in Mind
The following represents educational information rather than individual medical advice. Any decision to use NMN—especially if you have liver disease or other health conditions—should involve a qualified healthcare professional such as a registered dietitian or physician familiar with your medical history.
NMN supplementation may help improve insulin sensitivity, which is important for metabolic and liver health.
Liver-Focused Guidelines
Start low: Consider beginning at the lower end of commonly studied doses (125–250 mg/day) rather than immediately using 1,000 mg or more NMN. This allows you to assess tolerance while staying within well-studied ranges.
Check baseline labs: If feasible, work with a clinician to measure ALT, AST, ALP, GGT, and bilirubin before starting taking NMN supplements, especially if you drink alcohol regularly or use other medications.
Recheck periodically: For ongoing use exceeding 3 months, ask about rechecking liver enzymes every 3–6 months to detect subtle changes early.
Be cautious with alcohol: Avoid heavy drinking and binge patterns, which stress the liver and complicate interpretation of any abnormal labs.
Avoid “stacking” multiple NAD boosters: Combining NMN with high-dose niacin, nicotinamide riboside (NR), or other experimental compounds may increase unknown risks.
Stop and seek medical care if warning signs occur: Persistent right-upper-abdominal pain, dark urine, jaundice (yellowing of skin or eyes), severe fatigue, or unexplained nausea should prompt urgent medical evaluation.
Special Caution Groups
The following populations should exercise particular caution or consider avoidance:
Individuals with known chronic liver disease unless under specialist supervision
People undergoing active cancer treatment, given theoretical concerns about NAD+ and cell proliferation (including concerns related to Alzheimer’s disease research showing complex NAD+ relationships)
Pregnant or breastfeeding individuals, due to complete lack of safety data in these populations

How to Evaluate NMN Products From a Liver-Safety Perspective
Product quality can indirectly affect liver risk because contaminants, impurities, or mislabeling may pose far greater danger than NMN itself. Some products have been found to contain undeclared ingredients, heavy metals, or manufacturing residues that could stress the liver.
NMN is found naturally in foods such as broccoli, edamame, cucumbers, raw beef, certain vegetables, and cow’s milk, but in much smaller amounts than in supplements. This highlights the difference between obtaining NMN from natural dietary sources and the much higher doses present in supplements.
Criteria for Assessment
Third-party testing: Look for products tested by independent laboratories for purity and contaminants including heavy metals, solvents, and microbes. This represents a gold standard for supplement quality verification.
Clear labeling: The dose per capsule, total suggested daily doses, and full ingredient list (including excipients) should be transparent and easy to understand.
Reasonable dosing: Be wary of products promoting extremely high daily doses without referencing human studies or clinical research to support those recommendations.
Conservative claims: Prefer companies that do not make dramatic disease or cure claims. Overreaching marketing often indicates poor regulatory compliance and potentially questionable quality control.
The Mislabeling Problem
Misbranded NMN products have, in some markets, actually contained nicotinamide riboside (NR) or simple niacin instead of NMN. This complicates interpreting any anecdotal reports of side effects, including liver issues, because consumers may not actually be taking what they think they’re taking.
Cross-reference product information with published clinical doses (typically 250–600 mg/day) rather than relying on influencer anecdotes. NMN naturally occurs in small amounts in foods like raw beef, cow’s milk, and certain vegetables, but supplement doses far exceed what you’d obtain from foods alone.
Implications and Future Studies on NMN and Liver Safety
The implications of NMN supplementation for liver safety are far-reaching, especially as interest in its potential benefits for human health continues to grow. To fully understand the effects of NMN on liver health, future research must prioritize long-term, high-quality human clinical trials. These studies should include larger and more diverse populations, extended follow-up periods, and careful monitoring of liver function markers to capture both the benefits and any potential risks of NMN treatment.
Gold standard research methods—such as double-blind, placebo-controlled clinical trials—are essential to ensure reliable and unbiased results. In addition to assessing safety, future studies should explore the mechanisms by which NMN influences liver health, including its impact on insulin sensitivity, inflammation, and DNA repair. Understanding these pathways will help clarify how NMN supplementation can be optimized for maximum benefit and minimal risk.
By focusing on rigorous clinical research and prioritizing liver safety, the scientific community can unlock the full potential of NMN supplementation to improve human health and reduce the burden of age-related diseases. As new data emerge, ongoing evaluation and transparent communication will be key to ensuring that NMN remains a safe and effective option for those seeking to support their metabolic and liver health.
Key Takeaways: NMN and Liver Safety in 2026
Human clinical trials up to early 2026, mostly 8–24 weeks long and using 250–600 mg/day, have not shown a consistent signal of liver toxicity from NMN in generally healthy adults.
Routine liver markers (ALT, AST, ALP, GGT, bilirubin) have stayed within normal ranges for most participants, with adverse events typically mild and gastrointestinal in nature.
Animal studies and mechanistic research often suggest improved liver metabolic resilience with restored NAD+ levels, but these findings do not guarantee long-term safety in humans.
Major knowledge gaps remain for long-term use, higher doses, and people with existing liver disease, heavy alcohol intake, active cancer, or multiple medications.
Thoughtful dosing, medical guidance, periodic lab monitoring, and careful product selection can help reduce potential risks while taking NMN.
NMN is generally considered safe in short-term use at studied doses, but more research is needed for definitive long-term safety conclusions.
The effects of NMN on increased insulin sensitivity and blood pressure have shown promise in some studies, while research published in npj aging and other journals continues to expand our understanding. Placebo controlled trials using double blind methodology represent the most reliable source of safety information.
When weighing whether to try NMN, remember that supplements occupy a specific place in the health hierarchy. They’re additions to—not replacements for—fundamental liver-friendly habits. Limiting alcohol, maintaining a healthy weight through nutrition and regular exercise, managing stress, and getting adequate sleep form the foundation of liver health that no anti aging molecule can replace.
The science surrounding NMN continues to evolve rapidly, and what we understand in 2026 will likely expand significantly in the coming years. Approach NMN supplementation with appropriate caution, stay informed as new data emerge, and maintain open communication with your healthcare providers about any supplements you’re considering.

Further Reading
Explore more articles related to this topic:
- NMN and the Liver: Is it Safe or Toxic? (The Latest Data)
- The 2026 Runner’s Guide: How NMN Impacts VO2 Max and Aerobic Capacity
- NMN and Urolithin A: The New Frontier of Mitochondrial Health in 2026
- NMN for Brain Fog: How Boosting NAD+ Crosses the Blood-Brain Barrier
- NMN for Brain Fog: Exploring Its Role in Cognitive Function
- NMN and Alcohol: Does Drinking Deplete NAD+ and Negate Your Supplement?
- NMN Drug Interactions: Does NMN Interact with Statins, Blood Pressure Meds, and Antidepressants?